Abstract
Background
The Multicenter Selective Lymphadenectomy Trial-1 (MSLT-1) failed to demonstrate a survival advantage for sentinel lymph node biopsy (SNB) in melanoma. This may have been secondary to inadequate statistical power. This study was designed to determine the impact of SNB on melanoma-specific survival (MSS) in a larger patient cohort.
Methods
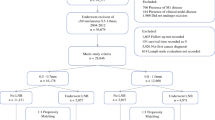
From 2003–2008, patients with tumors 1–4 mm in thickness and clinically negative nodes were identified within the SEER registry. Propensity score was used to create two matched cohorts: those who underwent a wide excision with SNB or wide excision alone.
Result
A total of 15,274 met inclusion criteria and 7,910 comprised the match cohort. Average age was 67.4 years. The majority were male (62.3 %) and white (97.2 %). Primary tumors were most frequently nonulcerated (77.1 %), located on the extremity (42.3 %), and T2 (64.1 %). There were 3,955 patients in both the SNB and observation groups. There was no significant difference in gender, ethnicity, ulceration status, primary site, or T-classification between the groups. Improved 5-year MSS was associated with SNB (85.7 vs. 84.0 %), female gender (88.3 vs. 82.7 %), absence of ulceration (87.5 vs. 75.7 %), extremity location (87.4 %), T2 disease (88.6 vs. 77.9 %), and a negative SNB (88.9 vs. 64.8 %). The relationships between observation [hazard ratio (HR) 1.18], male gender (HR 1.33), ulceration (HR 1.77), head-and-neck location (HR 1.34), and T3 disease (HR 1.82) persisted on multivariate analysis.
Conclusions
Status of the sentinel node is the strongest predictor of survival in patients with intermediate thickness melanoma. SNB compared with observation was associated with a modest survival advantage.

Similar content being viewed by others
References
Sim FH, Taylor WF, Ivins JC, Pritchard DJ, Soule EH. A prospective randomized study of the efficacy of routine elective lymphadenectomy in management of malignant melanoma. Preliminary results. Cancer. 1978;41(3):948–56.
Veronesi U, Adamus J, Bandiera DC, et al. Delayed regional lymph node dissection in stage I melanoma of the skin of the lower extremities. Cancer. 1982;49(11):2420–30.
Balch CM, Soong S, Ross MI, et al. Long-term results of a multi-institutional randomized trial comparing prognostic factors and surgical results for intermediate thickness melanomas (1.0 to 4.0 mm). Intergroup Melanoma Surgical Trial. Ann Surg Oncol. 2000;7(2):87–97.
Balch CM, Soong SJ, Bartolucci AA, et al. Efficacy of an elective regional lymph node dissection of 1 to 4 mm thick melanomas for patients 60 years of age and younger. Ann Surg. 1996;224(3):255–63; discussion 263–6.
Cascinelli N, Morabito A, Santinami M, MacKie RM, Belli F. Immediate or delayed dissection of regional nodes in patients with melanoma of the trunk: a randomised trial. WHO Melanoma Programme. Lancet. 1998;351(9105):793–6.
Wong JH, Cagle LA, Morton DL. Lymphatic drainage of skin to a sentinel lymph node in a feline model. Ann Surg. 1991;214(5):637–41.
Morton DL, Thompson JF, Cochran AJ, et al. Sentinel-node biopsy or nodal observation in melanoma. N Engl J Med. 2006;355(13):1307–17.
Program SEaERS. SEER Research Data (1973–2009). SEER*Stat 2011; http://seer.cancer.gov/about/overview.html.
Wright BE, Scheri RP, Ye X, et al. Importance of sentinel lymph node biopsy in patients with thin melanoma. Arch Surg. 2008;143(9):892–9; discussion 899–900.
Edge SB, Byrd DR, Compton CC, Fritz AG, Green FL, Trotti A. AJCC cancer staging manual, 7th ed. Philadelphia: Lippincott-Raven; 2010.
Rosenbaum PR. Observational studies, 2nd ed. New York: Springer-Verlag; 2002.
Perraillon MC. Matching with propensity scores to reduce bias in observational studies. NESUG. 2006:1–10.
D’Agostino RH. Propensity score methods for bias reduction in the comparison of a treatment of a non-randomized control group. Stat Med. 1998;17:2265–81.
Morton DL, Wen DR, Wong JH, et al. Technical details of intraoperative lymphatic mapping for early stage melanoma. Arch Surg. 1992;127(4):392–9.
Cascinelli N. WHO declares lymphatic mapping to be standard of care for melanoma. Oncology. 1999;13:288.
Ross MI, Gershenwald JE. How should we view the results of the Multicenter Selective Lymphadenectomy Trial-1 (MSLT-1)? Ann Surg Oncol. 2008;15(3):670–3.
Morton DL, Thompson JF, Cochran AJ, et al. Final trial report of sentinel-node biopsy versus nodal observation in melanoma. N Engl J Med. 2014;370(7):599–609.
Iwashyna TJL, Lamont EB. Effectiveness of adjuvant fluorouracil in clinical practice: a population-based cohort study of elderly patients with stage III colon cancer. J Clin Oncol. 2002;20:3992–8.
Sun M, Trinh QD, Bianchi M, et al. A non-cancer-related survival benefit is associated with partial nephrectomy. Eur Urol. 2012;61:725–731.
Abdollah F, Sun M, Schmitges J, et al. Cancer-specific and other-cause mortality after radical prostatectomy versus observation in patients with prostate cancer: competing-risks analysis of a large North American population-based cohort. Eur Urol. 2011;60:920–30.
Reeve BB, Smith AW, Arora NK, Hays RD. Reducing bias in cancer research: application of propensity score matching. Health Care Financ Rev. 2008;29(4):69–80.
Rubin DB. Estimating causal effects from large data sets using propensity scores. Ann Intern Med. 1997;127(8 Pt 2):757–63.
Nowecki ZI, Rutkowski P, Michej W. The survival benefit to patients with positive sentinel node melanoma after completion lymph node dissection may be limited to the subgroup with a primary lesion Breslow thickness greater than 1.0 and less than or equal to 4 mm (pT2–pT3). Ann Surg Oncol. 2008;15(8):2223–34.
Faries M. Survival and the sentinel lymph node in melanoma. Ann Surg Oncol. 2010;17:18–20.
Gershenwald JE, Thompson W, Mansfield PF, et al. Multi-institutional melanoma lymphatic mapping experience: the prognostic value of sentinel lymph node status in 612 stage I or II melanoma patients. J Clin Oncol. 1999;17(3):976–83.
Nowecki ZI, Rutkowski P, Nasierowska-Guttmejer A, Ruka W. Sentinel lymph node biopsy in melanoma patients with clinically negative regional lymph nodes—one institution’s experience. Melanoma Res. 2003;13(1):35–43.
Fadaki N, Li R, Parrett B, et al. Is head and neck melanoma different from trunk and extremity melanomas with respect to sentinel lymph node status and clinical outcome? Ann Surg Oncol. 2013;20(9):3089–97.
Chao C, Wong SL, Edwards MJ, et al. Sentinel lymph node biopsy for head and neck melanomas. Ann Surg Oncol. 2003;10(1):21–6.
Gimotty PA, Yoon F, Hammond R, Rosenbaum P, Guerry D. Therapeutic effect of sentinel lymph node biopsy in melanoma remains an open question. J Clin Oncol. 2009;27(26):4236–8.
Grob JJ, Dreno B, Salmoniere P, et al. Randomised trial of interferon alfa-2a as adjuvant therapy in resected primary melanoma thicker than 1.5 mm without clinically detectable node metastasis. Lancet. 1998;351:1905–10.
Kirkwood JM, Strawderman MH, Ernstoff MS, Smith TJ, Borden EC, Blum RH. Interferon alfa-2b adjuvant therapy of high-risk resected cutaneous melanoma: the Eastern Cooperative Oncology Group Trial EST 1684. J Clin Oncol. 1996;14(1):7–17.
Homsi J, Kashani-Sabet M, Messina JL, Daud A. Cutaneous melanoma: prognostic factors. Cancer Control. 2005;12(4):223–9.
Winkelmayer WC, Kurth T. Propensity scores: help or hype? Nephrol Dial Transplant. 2004;19(7):1671–73.
Kachare SD, Agle SC, Englert ZP, et al. Malignant blue nevus: clinicopathologically similar to melanoma. Am Surg. 2013;79(7):651–6.
Kachare SD, Wong JH, Vohra NA, Zervos EE, Fitzgerald TL. Sentinel lymph node biopsy is associated with improved survival in Merkel cell carcinoma. Ann Surg Oncol. 2014;21(5):1624–30.
Mojica P, Smith D, Ellenhorn JD. Adjuvant radiation therapy is associated with improved survival in Merkel cell carcinoma of the skin. J Clin Oncol. 2007;25(9):1043–7.
Torjesen I. Sentinel node biopsy for melanoma: unnecessary treatment? BMJ. 2013;8:1.
Institute JWC. Multicenter Selective Lymphadenectomy Trial II (MSLT-II). http://clinicaltrials.gov/show/NCT00297895, 2014.
Acknowledgment
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kachare, S.D., Brinkley, J., Wong, J.H. et al. The Influence of Sentinel Lymph Node Biopsy on Survival for Intermediate-Thickness Melanoma. Ann Surg Oncol 21, 3377–3385 (2014). https://doi.org/10.1245/s10434-014-3954-5
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-014-3954-5