Abstract
Background
Free peritoneal tumor cells (FPTCs) are an independent prognostic factor in patients undergoing curative resection for gastric carcinoma. Whether neoadjuvant chemotherapy (NAC) can eliminate FPTCs in the peritoneal lavage remains unclear. The aim of the study was to determine the effect of NAC on FPTCs.
Methods
From 1994 to 2000, data from a total of 61 patients with resectable gastric cancer were analyzed. Peritoneal cytology was performed before NAC at laparoscopy and at tumor resection. A minimum of 6 weeks of NAC, consisting of cisplatin, folinic acid, and fluorouracil, was administered. FPTCs were detected immunohistochemically with Ber-EP4 antibody.
Results
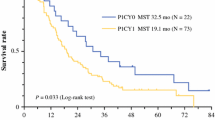
No FPTCs could be detected in 42 patients (69%), compared to 19 (31%) with FPTCs before NAC. During chemotherapy, 10 (24%) of 42 patients developed FPTCs, and 7 (37%) of 19 patients reverted from positive to negative. Patients who became FPTC negative (n = 7) showed an improved median survival (36.1 months) and a longer 2-year survival (71.4%) compared to FPTC-positive patients before and after NAC (n = 12), with a median survival of 9.2 months and a 2-year survival rate of 25%. In contrast, patients who reverted from FPTC negative to positive during NAC (n = 10) had a median survival of 18.5 months and a 2-year survival of only 20%. Multivariate analysis identified ypN category and FPTC change as independent prognostic factors.
Conclusions
NAC for patients with positive cytology could lead to FPTC negativity in a subset of patients and improve their prognosis. However, NAC might be a risky strategy for almost one-quarter of patients whose disease develops positive cytology.

Similar content being viewed by others
References
D’Angelica M, Gonen M, Brennan MF, et al. Patterns of initial recurrence in completely resected gastric adenocarcinoma. Ann Surg. 2004;240:808–16.
Allum WH, Powell DJ, McConkey CC, Fielding JW. Gastric cancer: a 25-year review. Br J Surg. 1989;76:535–40.
Siewert JR, Lordick F, Ott K, et al. Curative vs palliative strategies in locoregional recurrence of gastrointestinal malignancies. Chirurg. 2006;77:227–35.
Kodera Y, Nakanishi H, Yamamura Y, et al. Prognostic value and clinical implications of disseminated cancer cells in the peritoneal cavity detected by reverse transcriptase–polymerase chain reaction and cytology. Int J Cancer. 1998;79:429–33.
Ikeguchi M, Matsumoto S, Yoshioka S, et al. Laparoscopic-assisted intraperitoneal chemotherapy for patients with scirrhous gastric cancer. Chemotherapy. 2005;51:15–20.
Yoo CH, Noh SH, Shin DW, et al. Recurrence following curative resection for gastric carcinoma. Br J Surg. 2000;87:236–42.
Kitamura Y, Hayashi K, Sasagawa T, et al. Pilot study of S-1 in patients with disseminated gastric cancer. Drugs Exp Clin Res. 2003;29:125–30.
Nekarda H, Gess C, Stark M, et al. Immunocytochemically detected free peritoneal tumour cells (FPTC) are a strong prognostic factor in gastric carcinoma. Br J Cancer. 1999;79:611–9.
Katsuragi K, Yashiro M, Sawada T, et al. Prognostic impact of PCR-based identification of isolated tumour cells in the peritoneal lavage fluid of gastric cancer patients who underwent a curative R0 resection. Br J Cancer. 2007;97:550–6.
Feussner H, Hartl F. Staging laparoscopy in oncology. Chirurg. 2006;77:971–80.
Cunningham D, Allum WH, Stenning SP, et al. Perioperative chemotherapy versus surgery alone for resectable gastroesophageal cancer. N Engl J Med. 2006;355:11–20.
Schuhmacher C, Schlag P, Lordick F, Hohenberger W, et al. NAC versus surgery alone for locally advanced adenocarcinoma of the stomach and cardia: randomized EORTC phase III trial (abstract). J Clin Oncol. 2009;27(Suppl):4510
Boige V, Pignon J, Saint-Aubert B, Lasser P, et al. Final results of a randomized trial comparing preoperative 5-fluorouracil (F)/cisplatin (P) to surgery alone in adenocarcinoma of stomach and lower esophagus (ASLE): FNLCC ACCORD07-FFCD 9703 trial (abstract). J Clin Oncol. 2007;25(Suppl):4510
Ott K, Fink U, Becker K, et al. Prediction of response to preoperative chemotherapy in gastric carcinoma by metabolic imaging: results of a prospective trial. J Clin Oncol. 2003;21:4604–10.
Ott K, Herrmann K, Lordick F, et al. Early metabolic response evaluation by fluorine-18 fluorodeoxyglucose positron emission tomography allows in vivo testing of chemosensitivity in gastric cancer: long-term results of a prospective study. Clin Cancer Res. 2008;14:2012–8.
Beer TW, Shepherd P, Theaker JM. Ber EP4 and epithelial membrane antigen aid distinction of basal cell, squamous cell and basosquamous carcinomas of the skin. Histopathology. 2000;37:218–23.
Greene FL, Page DL, Fleming ID, Fritz A, Balch CM, editors. AJCC cancer staging manual. 6th ed. New York: Springer; 2002.
Becker K, Mueller JD, Schulmacher C, et al. Histomorphology and grading of regression in gastric carcinoma treated with NAC. Cancer. 2003;98:1521–30.
Japanese Research Society for Gastric Cancer. The general rules for the gastric cancer study. Tokyo: Kanahara Shuppan; 1995.
Okabe H, Ueda S, Obama K, et al. Induction chemotherapy with S-1 plus cisplatin followed by surgery for treatment of gastric cancer with peritoneal dissemination. Ann Surg Oncol. 2009;16:3227–36.
Yonemura Y, Bandou E, Sawa T, et al. Neoadjuvant treatment of gastric cancer with peritoneal dissemination. Eur J Surg Oncol. 2006;32:661–5.
Parsons S, Muarwa PX, Kolesnik OO. Treatment of epithelial cancer patients with malignant ascites using catumaxomab: results of the non-ovarian stratum of a phase II/III study (abstract). In: Proceedings of the Gastrointestinal Cancer Symposium; 2008. p. 102.
Boku T, Nakane Y, Minoura T, et al. Prognostic significance of serosal invasion and free intraperitoneal cancer cells in gastric cancer. Br J Surg. 1990;77:436–9.
Gretschel S, Siegel R, Estevez-Schwarz L, et al. Surgical strategies for gastric cancer with synchronous PC. Br J Surg. 2006;93:1530–5.
Suzuki T, Ochiai T, Hayashi H, et al. Peritoneal lavage cytology findings as prognostic factor for gastric cancer. Semin Surg Oncol. 1999;17:103–7.
Abe S, Yoshimura H, Tabara H, et al. Curative resection of gastric cancer: limitation of peritoneal lavage cytology in predicting the outcome. J Surg Oncol. 1995;59:226–9.
Author information
Authors and Affiliations
Corresponding author
Additional information
The first two authors contributed equally to this article.
Rights and permissions
About this article
Cite this article
Lorenzen, S., Panzram, B., Rosenberg, R. et al. Prognostic Significance of Free Peritoneal Tumor Cells in the Peritoneal Cavity Before and After Neoadjuvant Chemotherapy in Patients with Gastric Carcinoma Undergoing Potentially Curative Resection. Ann Surg Oncol 17, 2733–2739 (2010). https://doi.org/10.1245/s10434-010-1090-4
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-010-1090-4