Abstract
Background: Prostaglandins (PGs) were shown in vitro to suppress several functions of cellular immunity. It is unclear, however, whether physiological levels of PGs can suppress cellular immunity in vivo and whether such suppression would compromise postoperative host resistance to metastasis.
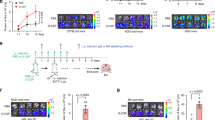
Methods: Fischer 344 rats were administered PGE2 in doses (18 to 300 μg/kg subcutaneously) that increased the serum levels approximately 2- to 4-fold. We then assessed the number and activity of circulating natural killer (NK) cells, as well as rats’ resistance to experimental metastasis of a syngeneic NK-sensitive tumor (MADB106). To study whether endogenously released PGs after surgery compromise these indices, we tested whether laparotomy adversely affects them and whether a cyclooxygenase-synthesis inhibitor, indomethacin (4 mg/kg), attenuates these effects.
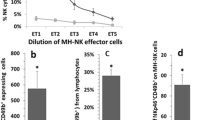
Results: PGE2 dose-dependently suppressed NK activity per NK cell and dose-dependently increased 4- and 24-hour MADB106 lung tumor retention (LTR); 240 μg/kg of PGE2 quadrupled the number of lung metastases counted 3 weeks later. Selective depletion of NK cells abrogated the promotion of LTR by PGE2. Surgery significantly suppressed NK activity and increased MADB106 LTR, and indomethacin halved these effects without affecting nonoperated rats.
Conclusions:PGE2 is a potent in vivo suppressor of NK activity, and its postoperative release may promote tumor recurrence.
Similar content being viewed by others
REFERENCES
Kim S, Iizuka K, Aguila HL, et al. In vivo natural killer cell activities revealed by natural killer cell- deficient mice. Proc Natl Acad Sci U S A 2000;97:2731–6.
Lanier LL. On guard—activating NK cell receptors. Nat Immunol 2001;2:23–7.
Ogawa K, Hirai M, Katsube T, et al. Suppression of cellular immunity by surgical stress. Surgery 2000;127:329–36.
Ben-Eliyahu S. The promotion of tumor metastasis by surgery and stress: immunological basis and implications for psychoneuroimmunology (PNI). Brain Behav Immun 2003;17:S27–S36.
Ben-Eliyahu S, Page GG, Yirmiya R, Shakhar G. Evidence that stress and surgical interventions promote tumor development by suppressing natural killer cell activity. Int J Cancer 1999;80:880–8.
Zoller M, Heumann U, Betzler M, et al. Depression of nonadaptive immunity after surgical stress: influence on metastatic spread. Invasion Metastasis 1989;9:46–68.
Toft P, Dagnaes-Hansen F, Tonnesen E, Basse PM. The effect of surgical stress and endotoxin-induced sepsis on the NK-cell activity, distribution and pulmonary clearance of YAC-1 and melanoma cells. APMIS 1999;107:359–64.
Da Costa ML, Redmond P, Bouchier-Hayes DJ. The effect of laparotomy and laparoscopy on the establishment of spontaneous tumor metastases. Surgery 1998;124:516–25.
Liedtke CM. Differentiated properties of rabbit tracheal epithelial cells in primary culture. Am J Physiol 1988;255(6 Pt 1):C760–70.
Ueda F, Ideguchi K, Taniguchi N, Kimura K. Adrenergic regulation of prostaglandin biosynthesis in cultured rabbit gastric epithelial cells. Jpn J Pharmacol 1994;65:113–20.
Baxevanis CN, Reclos GJ, Gritzapis AD, et al. Elevated prostaglandin E2 production by monocytes is responsible for the depressed levels of natural killer and lymphokine-activated killer cell function in patients with breast cancer. Cancer 1993;72:491–501.
Skibinski G, Kelly RW, Harrison CM, et al. Relative immunosuppressive activity of human seminal prostaglandins. J Reprod Immunol 1992;22:185–95.
Specht C, Bexten S, Kolsch E, Pauels HG. Prostaglandins, but not tumor-derived IL-10, shut down concomitant tumor-specific CTL responses during murine plasmacytoma progression. Int J Cancer 2001;91:705–12.
Goodwin JS, Ceuppens J. Regulation of the immune response by prostaglandins. J Clin Immunol 1983;3:295–315.
Uotila P. The role of cyclic AMP and oxygen intermediates in the inhibition of cellular immunity in cancer. Cancer Immunol Immunother 1996;43:1–9.
Leung KH, Koren HS. Regulation of human natural killing. III. Mechanism for interferon induction of loss of susceptibility to suppression by cyclic AMP elevating agents. J Immunol 1984;132:1445–50.
Rhind SG, Gannon GA, Suzui M, et al. Indomethacin inhibits circulating PGE2 and reverses postexercise suppression of natural killer cell activity. Am J Physiol 1999;276(5 Pt 2):R1496–505.
Nieman DC, Ahle JC, Henson DA, et al. Indomethacin does not alter natural killer cell response to 2.5 h of running. J Appl Physiol 1995;79:748–55.
Leung KH. Inhibition of human NK cell and LAK cell cytotoxicity and differentiation by PGE2. Cell Immunol 1989;123:384–95.
Garcia-Penarrubia P, Bankhurst AD, Koster FT. Prostaglandins from human T suppressor/cytotoxic cells modulate natural killer antibacterial activity. J Exp Med 1989;170:601–6.
Lauzon W, Lemaire I. Alveolar macrophage inhibition of lung-associated NK activity: involvement of prostaglandins and transforming growth factor-beta 1. Exp Lung Res 1994;20:331–49.
Goodwin JS, Webb DR. Regulation of the immune response by prostaglandins. Clin Immunol Immunopathol 1980;15:106–22.
Baxevanis CN, Papilas K, Dedoussis GV, et al. Abnormal cytokine serum levels correlate with impaired cellular immune responses after surgery. Clin Immunol Immunopathol 1994;71:82–8.
Vitoratos N, Hassiakos D, Louridas C, et al. Prostaglandin F1a and prostaglandin E2 plasma levels after transvaginal cervical cerclage. Clin Exp Obstet Gynecol 1996;23:21–5.
Brinkmann A, Seeling W, Wolf CF, et al. Ibuprofen does not impair renal function in patients undergoing infrarenal aortic surgery with epidural anaesthesia. Intensive Care Med 1998;24:322–8.
Okuno K, Jinnai H, Lee YS, et al. A high level of prostaglandin E2 (PGE2) in the portal vein suppresses liver-associated immunity and promotes liver metastases. Surg Today 1995;25:954–8.
Parsson HN, Lord RS, Scott K, Zemack G. Maintaining carotid flow by shunting during carotid endarterectomy diminishes the inflammatory response mediating ischaemic brain injury. Eur J Vasc Endovasc Surg 2000;19:124–30.
Bash JA, Vogel D. Cellular immunosenescence in F344 rats: decreased natural killer (NK) cell activity involves changes in regulatory interactions between NK cells, interferon, prostaglandin and macrophages. Mech Ageing Dev 1984;24:49–65.
Koren HS, Leung KH. Modulation of human NK cells by interferon and prostaglandin E2. Mol Immunol 1982;19:1341–6.
Ben-Eliyahu S, Shakhar G. The impact of stress, catecholamines, and the menstrual cycle on NK activity and tumor development: from in vitro studies to biological significance Ader R, Felten DL, Cohen N Eds. Psychoneuroimmunology, vol 2. San Diego:: Academic Press, 2001: 545–63.
Hellstrand K, Hermodsson S, Strannegard O. Evidence for a beta-adrenoceptor-mediated regulation of human natural killer cells. J Immunol 1985;134:4095–9.
Huribal M, Cunningham ME, D’Aiuto ML, et al. Endothelin-1 and prostaglandin E2 levels increase in patients with burns. J Am Coll Surg 1995;180:318–22.
Ben-Eliyahu S, Page GG, Shakhar G, Taylor AN. Increased susceptibility to metastasis during pro-oestrus/oestrus in rats: possible role of oestradiol and natural killer cells. Br J Cancer 1996;74:1900–7.
Page GG, Ben-Eliyahu S, Liebeskind JC. The role of LGL/NK cells in surgery-induced promotion of metastasis and its attenuation by morphine. Brain Behav Immun 1994;8:241–50.
Barlozzari T, Leonhardt J, Wiltrout RH, et al. Direct evidence for the role of LGL in the inhibition of experimental tumor metastases. J Immunol 1985;134:2783–9.
Barlozzari T, Reynolds CW, Herberman RB. In vivo role of natural killer cells: involvement of large granular lymphocytes in the clearance of tumor cells in anti-asialo GM1-treated rats. J Immunol 1983;131:1024–7.
Shakhar G, Ben-Eliyahu S. In vivo beta-adrenergic stimulation suppresses natural killer activity and compromises resistance to tumor metastasis in rats. J Immunol 1998;160:3251–8.
Page GG, Ben-Eliyahu S. A role for NK cells in greater susceptibility of young rats to metastatic formation. Dev Comp Immunol 1999;23:87–96.
Hodgson DM, Yirmiya R, Chiappelli F, Taylor AN. Intracerebral interleukin-1beta impairs response to tumor invasion: involvement of adrenal catecholamines. Brain Res 1999;816:200–8.
Yirmiya R, Ben-Eliyahu S, Gale RP, et al. Ethanol increases tumor progression in rats: possible involvement of natural killer cells. Brain Behav Immun 1992;6:74–86.
Chambers WH, Brumfield AM, Hanley-Yanez K, et al. Functional heterogeneity between NKR-P1bright/Lycopersicon esculentum lectin (L.E.)bright and NKR-P1bright/L.E.dim subpopulations of rat natural killer cells. J Immunol 1992;148:3658–65.
Chambers WH, Vujanovic NL, DeLeo AB, et al. Monoclonal antibody to a triggering structure expressed on rat natural killer cells and adherent lymphokine-activated killer cells. J Exp Med 1989;169:1373–89.
Ben-Eliyahu S, Page GG. In vivo assessment of natural killer cell activity in rats. Prog Neuroendocrineimmunol 1992;5:199–214.
Ben-Eliyahu S, Page GG, Yirmiya R, Taylor AN. Acute alcohol intoxication suppresses natural killer cell activity and promotes tumor metastasis. Nat Med 1996;2:457–60.
von Horsten S, Helfritz A, Kuhlmann S, et al. Stereological quantification of carboxyfluorescein-labeled rat lung metastasis: a new method for the assessment of natural killer cell activity and tumor adhesion in vivo and in situ. J Immunol Methods 2000;239:25–34.
Ben-Eliyahu S, Abudarham N, Shakhar G, et al. Protecting cellular immunity from suppression by surgery and stress hormones: an approach to prevent surgery-induced tumor progression (abstract). Brain Behav Immun 2002;16:176.
Shingu K, Helfritz A, Kuhlmann S, et al. Kinetics of the early recruitment of leukocyte subsets at the sites of tumor cells in the lungs: natural killer (NK) cells rapidly attract monocytes but not lymphocytes in the surveillance of micrometastasis. Int J Cancer 2002;99:74–81.
Kong LY, Luster MI, Dixon D, et al. Inhibition of lung immunity after intratracheal instillation of benzo(a)pyrene. Am J Respir Crit Care Med 1994;150:1123–9.
Ben-Eliyahu S, Shakhar G, Rosenne E, et al. Hypothermia in barbiturate-anesthetized rats suppresses natural killer cell activity and compromises resistance to tumor metastasis: a role for adrenergic mechanisms. Anesthesiology 1999;91:732–40.
Page GG, Ben Eliyahu S, Yirmiya R, Liebeskind JC. Morphine attenuates surgery-induced enhancement of metastatic colonization in rats. Pain 1993;54:21–8.
Bothwell W, Verburg M, Wynalda M, et al. A radioimmunoassay for the unstable pulmonary metabolites of prostaglandin E1 and E2: an indirect index of their in vivo disposition and pharmacokinetics. J Pharmacol Exp Ther 1982;220:229–35.
Huang M, Sharma S, Mao JT, Dubinett SM. Non-small cell lung cancer-derived soluble mediators and prostaglandin E2 enhance peripheral blood lymphocyte IL-10 transcription and protein production. J Immunol 1996;157:5512–20.
Chambrier C, Chassard D, Bienvenu J, et al. Cytokine and hormonal changes after cholecystectomy. Effect of ibuprofen pretreatment. Ann Surg 1996;224:178–82.
Wojtowicz-Praga S. Reversal of tumor-induced immunosuppression: a new approach to cancer therapy. J Immunother 1997;20:165–77.
Menetrier-Caux C, Bain C, Favrot MC, et al. Renal cell carcinoma induces interleukin 10 and prostaglandin E2 production by monocytes. Br J Cancer 1999;79:119–30.
Carter JJ. The immunologic consequences of laparoscopy in oncology. Surg Oncol Clin North Am 2001;10:655–77.
Brittenden J, Heys SD, Ross J, Eremin O. Natural killer cells and cancer. Cancer 1996;77:1226–43.
Andreesen R, Hennemann B, Krause SW. Adoptive immunotherapy of cancer using monocyte-derived macrophages: rationale, current status, and perspectives. J Leukoc Biol 1998;64:419–26.
Fidler IJ. Therapy of cancer metastasis by systemic activation of macrophages. Adv Pharmacol 1994;30:271–326.
Faist E, Schinkel C, Zimmer S. Update on the mechanisms of immune suppression of injury and immune modulation. World J Surg 1996;20:454–9.
Koda K, Saito N, Takiguchi N, et al. Preoperative natural killer cell activity: correlation with distant metastases in curatively research colorectal carcinomas. Int Surg 1997;82:190–3.
McCoy JL, Rucker R, Petros JA. Cell-mediated immunity to tumor-associated antigens is a better predictor of survival in early stage breast cancer than stage, grade or lymph node status. Breast Cancer Res Treat 2000;60:227–34.
Fujisawa T, Yamaguchi Y. Autologous tumor killing activity as a prognostic factor in primary resected nonsmall cell carcinoma of the lung. Cancer 1997;79:474–81.
Schantz SP, Brown BW, Lira E, et al. Evidence for the role of natural immunity in the control of metastatic spread of head and neck cancer. Cancer Immunol Immunother 1987;25:141–8.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yakar, I., Melamed, R., Shakhar, G. et al. Prostaglandin E2 Suppresses NK Activity In Vivo and Promotes Postoperative Tumor Metastasis in Rats. Ann Surg Oncol 10, 469–479 (2003). https://doi.org/10.1245/ASO.2003.08.017
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1245/ASO.2003.08.017