Key Points
-
Birt–Hogg–Dubé (BHD) syndrome is an autosomal dominant inherited renal cancer disorder that predisposes at-risk individuals to benign, cutaneous fibrofolliculomas, pulmonary cysts, spontaneous pneumothoraces and increased risk of renal neoplasia
-
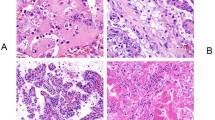
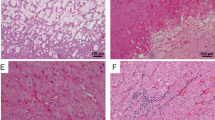
Renal tumours that develop in the setting of BHD syndrome are most often bilateral, multifocal hybrid oncocytic tumours and chromophobe renal cell carcinomas, but patients can present with other histologies
-
Germline mutations in FLCN, predicted to prematurely truncate the protein, predispose to BHD syndrome; renal tumours show somatic inactivation or loss of the remaining FLCN allele, confirming a tumour suppressor function
-
FLCN interacts with the novel proteins FNIP1 and FNIP2, as well as AMPK, a negative regulator of mTOR, and acts to modulate the AKT–TOR pathway
-
Other pathways in which FLCN might have a role include regulation of TFE3 and TFEB transcriptional activity, amino-acid-dependent mTOR activation through Rag GTPases, TGFβ signalling, PGC1α-driven mitochondrial biogenesis, and autophagy
-
Surgery is currently the only available therapy for BHD-associated renal tumours, but elucidation of FLCN-interacting pathways, deregulated in FLCN-deficient renal cancer, will hopefully enable the development of effective targeted therapies
Abstract
Birt–Hogg–Dubé (BHD) syndrome is an inherited renal cancer syndrome in which affected individuals are at risk of developing benign cutaneous fibrofolliculomas, bilateral pulmonary cysts and spontaneous pneumothoraces, and kidney tumours. Bilateral multifocal renal tumours that develop in BHD syndrome are most frequently hybrid oncocytic tumours and chromophobe renal carcinoma, but can present with other histologies. Germline mutations in the FLCN gene on chromosome 17 are responsible for BHD syndrome—BHD-associated renal tumours display inactivation of the wild-type FLCN allele by somatic mutation or chromosomal loss, confirming that FLCN is a tumour suppressor gene that fits the classic two-hit model. FLCN interacts with two novel proteins, FNIP1 and FNIP2, and with AMPK, a negative regulator of mTOR. Studies with FLCN-deficient cell and animal models support a role for FLCN in modulating the AKT–mTOR pathway. Emerging evidence links FLCN with a number of other molecular pathways and cellular processes important for cell homeostasis that are frequently deregulated in cancer, including regulation of TFE3 and/or TFEB transcriptional activity, amino-acid-dependent mTOR activation through Rag GTPases, TGFβ signalling, PGC1α-driven mitochondrial biogenesis, and autophagy. Currently, surgical intervention is the only therapy available for BHD-associated renal tumours, but improved understanding of the FLCN pathway will hopefully lead to the development of effective forms of targeted systemic therapy for this disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Linehan, W. M., Srinivasan, R. & Schmidt, L. S. The genetic basis of kidney cancer: a metabolic disease. Nat. Rev. Urol. 7, 277–285 (2010).
Latif, F. et al. Identification of the von Hippel-Lindau disease tumor suppressor gene. Science 260, 1317–1320 (1993).
Cancer Genome Atlas Research Network. Comprehensive molecular characterization of clear cell renal cell carcinoma. Nature 499, 43–49 (2013).
Nickerson, M. L. et al. Improved identification of von Hippel-Lindau gene alterations in clear cell renal tumors. Clin. Cancer Res. 14, 4726–4734 (2008).
Schmidt, L. et al. Germline and somatic mutations in the tyrosine kinase domain of the MET proto-oncogene in papillary renal carcinomas. Nat. Genet. 16, 68–73 (1997).
Tomlinson, I.P. et al. Germline mutations in FH predispose to dominantly inherited uterine fibroids, skin leiomyomata and papillary renal cell cancer. Nat. Genet. 30, 406–410 (2002).
Nickerson, M. L. et al. Mutations in a novel gene lead to kidney tumors, lung wall defects, and benign tumors of the hair follicle in patients with the Birt-Hogg-Dubé syndrome. Cancer Cell 2, 157–164 (2002).
Birt, A. R., Hogg, G. R. & Dubé, W. J. Hereditary multiple fibrofolliculomas with trichodiscomas and acrochordons. Arch. Dermatol. 113, 1674–1677 (1977).
Pinkus, H., Coskey, R. J. & Burgess, G. A. Trichodiscoma: A benign tumor relating to haarscheibe (hair disc). J. Invest. Dermatol. 63, 212 (1974).
Hornstein, O. P. & Knickenberg, M. Perifollicular fibromatosis cutis with polyps of the colon—a cutaneo-intestinal syndrome sui generis. Arch. Dermatol. Res. 253, 161–175 (1975).
Binet, O., Robin, J., Vicart, M., Ventura, G. & Beltzer-Garelly, E. Fibromes perifolliculaires polypose colique familiale pneumothorax spontanes familiaux. Ann. Dermatol. Venereo. 113, 928–930 (1986).
Chung, J. Y., Ramos-Caro, F. A., Beers, B., Ford, M. J. & Flowers, F. Multiple lipomas, angiolipomas, and parathyroid adenomas in a patient with Birt-Hogg-Dubé syndrome. Int. J. Dermatol. 35, 365–367 (1996).
Toro, J. R. et al. Birt-Hogg-Dubé syndrome: a novel marker of kidney neoplasia. Arch. Dermatol. 135, 1195–1202 (1999).
Roth, J. S., Rabinowitz, A. D., Benson, M. & Grossman, M. E. Bilateral renal cell carcinoma in the Birt-Hogg-Dubé syndrome. J. Am. Acad. Dermatol. 29, 1055–1056 (1993).
Zbar, B. et al. Risk of renal and colonic neoplasms and spontaneous pneumothorax in the Birt-Hogg-Dubé syndrome. Cancer Epidemiol. Biomarkers Prev. 11, 393–400 (2002).
Schmidt, L. S. et al. Germline BHD-mutation spectrum and phenotype analysis of a large cohort of families with Birt-Hogg-Dubé syndrome. Am. J. Hum. Genet. 76, 1023–1033 (2005).
Toro, J. R. et al. BHD mutations, clinical and molecular genetic investigations of Birt-Hogg-Dubé syndrome: a new series of 50 families and a review of published reports. J. Med. Genet. 45, 321–331 (2008).
Leter, E. M. et al. Birt-Hogg-Dubé syndrome: clinical and genetic studies of 20 families. J. Invest. Dermatol. 128, 45–49 (2008).
Kluger, N. et al. Birt-Hogg-Dubé syndrome: clinical and genetic studies of 10 French families. Br. J. Dermatol. 162, 527–537 (2010).
Vernooij, M., Claessens, T., Luijten, M., van Steensel, M. A. & Coull, B. J. Birt-Hogg-Dubé syndrome and the skin. Fam. Cancer 12, 381–385 (2013).
Schulz, T. & Hartschuh, W. Birt-Hogg-Dubé syndrome and Hornstein-Knickenberg syndrome are the same. Different sectioning technique as the cause of different histology. J. Cutan. Pathol. 26, 55–61 (1999).
Kunogi, M. et al. Clinical and genetic spectrum of Birt-Hogg-Dubé syndrome patients in whom pneumothorax and/or multiple lung cysts are the presenting feature. J. Med. Genet. 47, 281–287 (2010).
Gunji, Y. et al. Mutations of the Birt-Hogg-Dubé´ gene in patients with multiple lung cysts and recurrent pneumothorax. J. Med. Genet. 44, 588–593 (2007).
Toro, J. R. et al. Lung cysts, spontaneous pneumothorax, and genetic associations in 89 families with Birt-Hogg-Dubé syndrome. Am. J. Respir. Crit. Care Med. 175, 1044–1053 (2007).
Ayo, D. S., Aughenbaugh, G. L., Yi, E. S., Hand, J. L. & Ryu, J. H. Cystic lung disease in Birt-Hogg-Dubé´syndrome. Chest 132, 679–684 (2007).
Gupta, N., Seyama, K. & McCormack, F. X. Pulmonary manifestations of Birt-Hogg-Dubé syndrome. Fam. Cancer 12, 387–396 (2013).
Tobino, K. et al. Characteristics of pulmonary cysts in Birt-Hogg-Dubé syndrome: thin-section CT findings of the chest in 12 patients. Eur. J. Radiol. 77, 403–409 (2011).
Bessis, D., Giraud, S. & Richard, S. A novel familial germline mutation in the initiator codon of the BHD gene in a patient with Birt-Hogg-Dubé syndrome. Br. J. Dermatol. 155, 1067–1069 (2006).
Houweling, A. C. et al. Renal cancer and pneumothorax risk in Birt-Hogg-Dubé syndrome; an analysis of 115 FLCN mutation carriers from 35 BHD families. Br. J. Cancer 105, 1912–1919 (2011).
Benusiglio, P. R. et al. Renal cell tumour characteristics in patients with the Birt-Hogg-Dubé cancer susceptibility syndrome: a retrospective, multicentre study. Orphanet J. Rare Dis. 9, 163 (2014).
Khoo, S. K. et al. Clinical and genetic studies of Birt-Hogg-Dubé syndrome. J. Med. Genet. 39, 906e12 (2002).
Pavlovich, C. P. et al. Renal tumors in the Birt-Hogg-Dubé syndrome. Am. J. Surg. Pathol. 26, 1542–1552 (2002).
Pavlovich, C. P. et al. Evaluation and management of renal tumors in the Birt-Hogg-Dubé syndrome. J. Urol. 173, 1482–1486 (2005).
Adamy, A. et al. Renal oncocytosis: management and clinical outcomes. J. Urol. 185, 795–801 (2011).
Tickoo, S. K. et al. Renal oncocytosis: A morphologic study of fourteen cases. Am. J. Surg. Pathol. 23, 1094–1101 (1999).
Liu, V., Kwan, T., Page, E. H. Parotid oncocytoma in the Birt-Hogg-Dubé syndrome. J. Am. Acad. Dermatol. 43, 1120–1122 (2000).
Maffé, A. et al. Constitutional FLCN mutations in patients with suspected Birt-Hogg-Dubé syndrome ascertained for non-cutaneous manifestations. Clin. Genet. 79, 345–354 (2011).
Pradella, L. M. et al. Where Birt-Hogg-Dubé meets Cowden syndrome: mirrored genetic defects in two cases of syndromic oncocytic tumours. Eur. J. Hum. Genet. 21, 1169–1172 (2013).
Rongioletti, F., Hazini, R., Gianotti, G. & Rebora, A. Fibrofolliculomas, trichodiscomas and achrocordons (Birt-Hogg-Dubé) associated with intestinal polyposis. Clin. Exp. Dermatol. 14, 72–74 (1989).
Le Guyadec, T. et al. Multiple trichodiscomas associated with colonic polyposis. Ann. Dermatol. Venereol. 125, 717–719 (1998).
Nahorski, M. S. et al. Investigation of the Birt-Hogg-Dubé´ tumour suppressor gene (FLCN) in familial and sporadic colorectal cancer. J. Med. Genet. 47, 385–390 (2010).
Weirich, G. et al. Familial renal oncocytoma: clinicopathological study of 5 families. J. Urol. 160, 335–340 (1998).
Schmidt, L. S. et al. Birt-Hogg-Dubé syndrome, a genodermatosis associated with spontaneous pneumothorax and kidney neoplasia, maps to chromosome 17p11.2. Am. J. Hum. Genet. 69, 876–882 (2001).
Khoo, S. K. et al. Birt-Hogg-Dubé syndrome: mapping of a novel hereditary neoplasia gene to chromosome 17p12q11.2. Oncogene 20, 5239–5242 (2001).
Lim, D. H. et al. A new locus-specific database (LSDB) for mutations in the folliculin (FLCN) gene. Hum. Mutat. 31, E1043–1051 (2010).
Leiden Open Variation Database. European Birt-Hogg-Dubé Consortium LOVD Gene homepage for FLCN [online], (2015).
Benhammou, J. N. et al. Identification of intragenic deletions and duplication in the FLCN gene in Birt-Hogg-Dubé syndrome. Genes Chromosomes Cancer 50, 466–477 (2011).
Sempau, L., Ruiz, I., González-Morán, A., Susanna, X. & Hansen, T. V. New mutation in the Birt Hogg Dubé gene. Actas Dermosifiliogr. 101, 637–640 (2010).
Nahorski, M. S. et al. Birt Hogg-Dubé syndrome-associated FLCN mutations disrupt protein stability. Hum. Mutat. 32, 921–929 (2011).
Shin, J.H. et al. Mutations of the Birt Hogg Dubé (BHD) gene in sporadic colorectal carcinomas and colorectal carcinoma cell lines with microsatellite instability. J. Med. Genet. 40, 364–367 (2003).
Knudson, A. G. Jr. Mutation and cancer: statistical study of retinoblastoma. Proc. Natl Acad. Sci. USA 68, 820–823 (1971).
Vocke, C. D. et al. High frequency of somatic frameshift BHD gene mutations in Birt-Hogg-Dubé-associated renal tumors. J. Natl Cancer Inst. 97, 931–935 (2005).
Lingaas, F. et al. A mutation in the canine BHD gene is associated with hereditary multifocal renal cystadenocarcinoma and nodular dermatofibrosis in the German shepherd dog. Hum. Mol. Genet. 12, 3043–3053 (2003).
Okimoto, K. et al. A germ-line insertion in the Birt Hogg Dubé (BHD) gene gives rise to the Nihon rat model of inherited renal cancer. Proc. Natl Acad. Sci. USA 101, 2023–2027 (2004).
Yang, Y. et al. The UOK 257 cell line: a novel model for studies of the human Birt-Hogg-Dubé gene pathway. Cancer Genet. Cytogenet. 180, 100–109 (2008).
Hong, S. B. et al. Tumor suppressor FLCN inhibits tumorigenesis of a FLCN-null renal cancer cell line and regulates expression of key molecules in TGF-beta signaling. Mol. Cancer 9, 160 (2010).
Hudon, V. et al. Renal tumour suppressor function of the Birt-Hogg-Dubé syndrome gene product folliculin. J. Med. Genet. 47, 182–189 (2010).
Sato, Y. et al. Integrated molecular analysis of clear-cell renal cell carcinoma. Nat. Genet. 45, 860–867 (2013).
Gad, S. et al. Mutations in BHD and TP53 genes, but not in HNF1beta gene, in a large series of sporadic chromophobe renal cell carcinoma. Br. J. Cancer 96, 336–340 (2007).
Khoo, S. K. et al. Inactivation of BHD in sporadic renal tumors. Cancer Res. 63, 4583–4587 (2003).
Fernandez da Silva, N. et al. Analysis of the Birt-Hogg-Dubé (BHD) tumour suppressor gene in sporadic renal cell carcinoma and colorectal cancer. J. Med. Genet. 40, 820–824 (2003).
Nagy, A., Zoubakov, D., Stupar, Z. & Kovacs, G. Lack of mutation of the folliculin gene in sporadic chromophobe renal cell carcinoma and renal oncocytoma. Int. J. Cancer 109, 472–475 (2004).
Davis, C. F. et al. The Cancer Genome Atlas Research Network. The somatic genomic landscape of chromophobe renal cell carcinoma. Cancer Cell 26, 319–330 (2014).
Baba, M. et al. Folliculin encoded by the BHD gene interacts with a binding protein, FNIP1, and AMPK, and is involved in AMPK and mTOR signaling. Proc. Natl Acad. Sci. USA 103, 15552–15557 (2006).
Shaw, R. J. LKB1 and AMP-activated protein kinase control of mTOR signalling and growth. Acta Physiol. (Oxf.) 196, 65–80 (2009).
Wang, L. et al. Serine 62 is a phosphorylation site in folliculin, the Birt-Hogg-Dubé gene product. FEBS Lett. 584, 39–43 (2010).
Hasumi, H. et al. Identification and characterization of a novel folliculin-interacting protein FNIP2. Gene 415, 60–67 (2008).
Takagi, Y. et al. Interaction of folliculin (Birt-Hogg-Dubé gene product) with a novel Fnip1-like (FnipL/Fnip2) protein. Oncogene 27, 5339–5347 (2008).
Komori, K. et al. Anovel protein, MAPO1, that functions in apoptosis triggered by O6-methylguanine mispair in DNA. Oncogene 28, 1142–1150 (2009).
Baba, M. et al. The Folliculin-FNIP1 pathway deleted in human Birt-Hogg-Dubé syndrome is required for mouse B cell development. Blood 120, 1254–1261 (2012).
Hasumi, H. et al. Folliculin-interacting proteins Fnip1 and Fnip2 play critical roles in kidney tumor suppression in cooperation with Flcn. Proc. Natl Acad. Sci. USA 112, E1624–E1631 (2015).
Park, H. et al. Disruption of Fnip1 reveals a metabolic checkpoint controlling B lymphocyte development. Immunity 36, 769–781 (2012).
Baba, M. et al. Kidney-targeted Birt-Hogg-Dubé gene inactivation in a mouse model: Erk1/2 and Akt-mTOR activation, cell hyperproliferation, and polycystic kidneys. J. Natl Cancer Inst. 100, 140–154 (2008).
Chen, J. et al. Deficiency of FLCN in mouse kidney led to development of polycystic kidneys and renal neoplasia. PLoS ONE 3, e3581 (2008).
Hasumi, Y. et al. Homozygous loss of BHD causes early embryonic lethality and kidney tumor development with activation of mTORC1 and mTORC2. Proc. Natl Acad. Sci. USA 106, 18722–18727 (2009).
Hartman, T. R. et al. The role of the Birt-Hogg-Dubé protein in mTOR activation and renal tumorigenesis. Oncogene 28, 1594–1604 (2009).
van Slegtenhorst, M. et al. The Birt-Hogg-Dubé and tuberous sclerosis complex homologs have opposing roles in amino acid homeostasis in Schizosaccharomyces pombe. J. Biol. Chem. 282, 24583–24590 (2007).
Bar-Peled, L. & Sabatini, D. M. Regulation of mTORC1 by amino acids. Trends Cell Biol. 24, 400–406 (2014).
Tsun, Z. Y. et al. The folliculin tumor suppressor is a GAP for the RagC/D GTPases that signal amino acid levels to mTORC1. Mol. Cell 52, 495–505 (2013).
Petit, C. S., Roczniak-Ferguson, A. & Ferguson, S. M. Recruitment of folliculin to lysosomes supports the amino acid-dependent activation of Rag GTPases. J. Cell Biol. 202, 1107–1122 (2013).
Nookala, R. K. et al. Crystal structure of folliculin reveals a hidDENN function in genetically inherited renal cancer. Open Biol. 2, 120071 (2012).
Hong, S. B. et al. Inactivation of the FLCN tumor suppressor gene induces TFE3 transcriptional activity by increasing its nuclear localization. PLoS ONE 5, e15793 (2010).
Roczniak-Ferguson, A. et al. The transcription factor TFEB links mTORC1 signaling to transcriptional control of lysosome homeostasis. Sci. Signal. 5, ra42 (2012).
Betschinger, J. et al. Exit from pluripotency is gated by intracellular redistribution of the bHLH transcription factor Tfe3. Cell 153, 335–347 (2013).
Wolf, A. et al. The armadillo protein p0071 regulates Rho signalling during cytokinesis. Nat. Cell Biol. 8, 1432–1440 (2006).
Medvetz, D. A. et al. Folliculin, the product of the Birt-Hogg-Dubé tumor suppressor gene, interacts with the adherens junction protein p0071 to regulate cell–cell adhesion. PLoS ONE 7, e47842 (2012).
Nahorski, M. S. et al. Folliculin interacts with p0071 (plakophilin4) and deficiency is associated with disordered RhoA signalling, epithelial polarization and cytokinesis. Hum. Mol. Genet. 21, 5268–5279 (2012).
Khabibullin, D. et al. Folliculin regulates cell-cell adhesion, AMPK, and mTORC1 in a celltypespecific manner in lung-derived cells. Physiol. Rep. 2, e12107 (2014).
Goncharova, E. A. et al. Folliculin controls lung alveolar enlargement and epithelial cell survival through Ecadherin, LKB1 and AMPK. Cell Rep. 7, 412–423 (2014).
Cash, T. P., Gruber, J. J., Hartman, T. R., Henske, E. P. & Simon, M. C. Loss of the Birt-Hogg-Dubé tumor suppressor results in apoptotic resistance due to aberrant TGFβ-mediated transcription. Oncogene 30, 2534–2546 (2011).
Possik, E. et al. Folliculin regulates ampk-dependent autophagy and metabolic stress survival. PLoS Genet. 10, e1004273 (2014).
Dunlop, E. A. et al. FLCN, a novel autophagy component, interacts with GABARAP and is regulated by ULK1 phosphorylation. Autophagy 10, 1749–1760 (2014).
Stamatakis, L., Metwalli, A. R., Middelton, L. A. & Linehan, W. M. Diagnosis and management of BHD-associated kidney cancer. Fam. Cancer 12, 397–402 (2013).
Menko, F. H. et al. European BHD Consortium. Birt-Hogg-Dubé syndrome: diagnosis and management. Lancet Oncol. 10, 1199–1206 (2009).
Jamis-Dow, C. A. et al. Small (≤3 cm) renal masses: detection with CT versus US and pathologic correlation. Radiology 198, 785–788 (1996).
Waldert, M. et al. Hybrid renal cell carcinomas containing histopathologic features of chromophobe renal cell carcinomas and oncocytomas have excellent oncologic outcomes. Eur. Urol. 57, 661–665 (2010).
Boris, R. S. et al. The impact of germline BHD mutation on histological concordance and clinical treatment of patients with bilateral renal masses and known unilateral oncocytoma. J. Urol. 185, 2050–2055 (2011).
Hes, O., Petersson, F., Kuroda, N., Hora, M. & Michal, M. Renal hybrid oncocytic/ chromophobe tumors—a review. Histol. Histopathol. 28, 1257–1264 (2013).
Kowalczyk, K. J. et al. Partial nephrectomy after previous radio frequency ablation: the National Cancer Institute experience. J. Urol. 182, 2158–2163 (2009).
Wile, G. E., Leyendecker, J. R., Krehbiel, K. A., Dyer, R. B. & Zagoria, R. J. CT and MR imaging after imaging-guided thermal ablation of renal neoplasms. Radiographics 27, 325–339 (2007).
Furuya, M. et al. Distinctive expression patterns of glycoprotein non-metastatic B and folliculin in renal tumors in patients with Birt-Hogg-Dubé syndrome. Cancer Sci. 106, 315–23 (2015).
Tickoo, S. K. et al. Ultrastructural observations on mitochondria and microvesicles in renal oncocytoma, chromophobe renal cell carcinoma, and eosinophilic variant of conventional (clear cell) renal cell carcinoma. Am. J. Surg. Pathol. 24, 1247–1256 (2000).
Gasparre, G. et al. Clonal expansion of mutated mitochondrial DNA is associated with tumor formation and complex I deficiency in the benign renal oncocytoma. Hum. Mol. Genet. 17, 986–995 (2008).
Klomp, J. A. et al. Birt-Hogg-Dubé renal tumors are genetically distinct from other renal neoplasias and are associated with up-regulation of mitochondrial gene expression. BMC Med. Genomics 3, 59 (2010).
Hasumi, H. et al. Regulation of mitochondrial oxidative metabolism by tumor suppressor FLCN. J. Natl Cancer Inst. 104, 1750–1764 (2012).
Hasumi, Y. et al. Folliculin (Flcn) inactivation leads to murine cardiac hypertrophy through mTORC1 deregulation. Hum. Mol. Genet. 23, 5706–5719 (2014).
Shuch, B. et al. Targeting the mTOR pathway in Chromophobe Kidney Cancer. J. Cancer 3, 152–157 (2012).
Nakamura, M. et al. A case of metastatic renal cell carcinoma associated with Birt-Hogg-Dubé syndrome treated with molecular-targeting agents [Japanese]. Hinyokika Kiyo 59, 503–506 (2013).
Lu, X. et al. Therapeutic targeting the loss of the Birt-Hogg-Dubé suppressor gene. Mol. Cancer Ther. 10, 80–89 (2011).
Lu, X. et al. Knockdown of Slingshot 2 (SSH2) serine phosphatase induces Caspase3 activation in human carcinoma cell lines with the loss of the Birt-Hogg-Dubé tumour suppressor gene (FLCN). Oncogene 33, 956–965 (2014).
Preston, R. S. et al. Absence of the Birt-Hogg-Dubé gene product is associated with increased hypoxia-inducible factor transcriptional activity and a loss of metabolic flexibility. Oncogene 30, 1159–1173 (2011).
Yan, M. et al. The tumor suppressor folliculin regulates AMPK-dependent metabolic transformation. J. Clin. Invest. 124, 2640–2650 (2014).
Murakami, T. et al. Identification and characterization of Birt-Hogg-Dubé-associated renal carcinoma. J. Pathol. 211, 524–531 (2007).
Acknowledgements
This work was supported by the Intramural Research Program of the National Institutes of Health (NIH), National Cancer Institute (NCI), Centre for Cancer Research. This project has been funded in part with Federal funds from the Frederick National Laboratory for Cancer Research, NIH, under Contract HHSN261200800001E (L.S.S.). The content of this publication does not necessarily reflect the views or policies of the Department of Health and Human Services, nor does mention of trade names, commercial products or organizations imply endorsements by the US Government. L.S.S. is an employee of Basic Research Program, Leidos Biomedical Research, Inc., Frederick National Laboratory for Cancer Research, Frederick, MD 21702.
Author information
Authors and Affiliations
Contributions
Both authors contributed to researching data for article and made substantial contributions to discussion of content, as well as writing and review/editing of the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Schmidt, L., Linehan, W. Molecular genetics and clinical features of Birt–Hogg–Dubé syndrome. Nat Rev Urol 12, 558–569 (2015). https://doi.org/10.1038/nrurol.2015.206
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrurol.2015.206
This article is cited by
-
Lysosomes as coordinators of cellular catabolism, metabolic signalling and organ physiology
Nature Reviews Molecular Cell Biology (2024)
-
Coexistent Sjogren’s syndrome and Birt-Hogg-Dube´ syndrome: a case report
BMC Pulmonary Medicine (2023)
-
Exons 1–3 deletion in FLCN is associated with increased risk of pneumothorax in Chinese patients with Birt-Hogg-Dubé syndrome
Orphanet Journal of Rare Diseases (2023)
-
Familial multiple discoid fibromas is linked to a locus on chromosome 5 including the FNIP1 gene
Journal of Human Genetics (2023)
-
Aspergillus tracheobronchitis with Birt-Hogg-Dubè syndrome as a rare cause of chronic cough
BMC Pulmonary Medicine (2022)