Abstract
Diffuse-type gastric carcinoma (DGC) is characterized by a highly malignant phenotype with prominent infiltration and stromal induction. We performed whole-exome sequencing on 30 DGC cases and found recurrent RHOA nonsynonymous mutations. With validation sequencing of an additional 57 cases, RHOA mutation was observed in 25.3% (22/87) of DGCs, with mutational hotspots affecting the Tyr42, Arg5 and Gly17 residues in RHOA protein. These positions are highly conserved among RHO family members, and Tyr42 and Arg5 are located outside the guanine nucleotide–binding pocket. Several lines of functional evidence indicated that mutant RHOA works in a gain-of-function manner. Comparison of mutational profiles for the major gastric cancer subtypes showed that RHOA mutations occur specifically in DGCs, the majority of which were histopathologically characterized by the presence of poorly differentiated adenocarcinomas together with more differentiated components in the gastric mucosa. Our findings identify a potential therapeutic target for this poor-prognosis subtype of gastric cancer with no available molecularly targeted drugs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Accession codes
References
Hohenberger, P. & Gretschel, S. Gastric cancer. Lancet 362, 305–315 (2003).
Guggenheim, D.E. & Shah, M.A. Gastric cancer epidemiology and risk factors. J. Surg. Oncol. 107, 230–236 (2013).
Kong, X., Wang, J.L., Chen, H.M. & Fang, J.Y. Comparison of the clinicopathological characteristics of young and elderly patients with gastric carcinoma: a meta analysis. J. Surg. Oncol. 106, 346–352 (2012).
Warneke, V.S. et al. Cohort study based on the seventh edition of the TNM classification for gastric cancer: proposal of a new staging system. J. Clin. Oncol. 29, 2364–2371 (2011).
Chiaravalli, A.M. et al. Histotype-based prognostic classification of gastric cancer. World J. Gastroenterol. 18, 896–904 (2012).
Stiekema, J. et al. Surgical treatment results of intestinal and diffuse type gastric cancer. Implications for a differentiated therapeutic approach? Eur. J. Surg. Oncol. 39, 686–693 (2013).
Tanner, M. et al. Amplification of HER-2 in gastric carcinoma: association with Topoisomerase IIα gene amplification, intestinal type, poor prognosis and sensitivity to trastuzumab. Ann. Oncol. 16, 273–278 (2005).
Bang, Y. et al. Pathological features of advanced gastric cancer (GC): relationship to human epidermal growth factor receptor 2 (HER2) positivity in the global screening programme of the ToGA trial. J. Clin. Oncol. 27, 4556 (2009).
Wang, K. et al. Exome sequencing identifies frequent mutation of ARID1A in molecular subtypes of gastric cancer. Nat. Genet. 43, 1219–1223 (2011).
Zang, Z.J. et al. Exome sequencing of gastric adenocarcinoma identifies recurrent somatic mutations in cell adhesion and chromatin remodeling genes. Nat. Genet. 44, 570–574 (2012).
Alexandrov, L.B. et al. Signatures of mutational processes in human cancer. Nature 500, 415–421 (2013).
Guilford, P. et al. E-cadherin germline mutations in familial gastric cancer. Nature 392, 402–405 (1998).
Chen, H.J. et al. The role of microtubule actin cross-linking factor 1 (MACF1) in the Wnt signaling pathway. Genes Dev. 20, 1933–1945 (2006).
Salinas, P.C. Modulation of the microtubule cytoskeleton: a role for a divergent canonical Wnt pathway. Trends Cell Biol. 17, 333–342 (2007).
Anastasiadis, P.Z. et al. Inhibition of RhoA by p120 catenin. Nat. Cell Biol. 2, 637–644 (2000).
Carothers, A.M. et al. Deficient E-cadherin adhesion in C57BL/6J-Min/+ mice is associated with increased tyrosine kinase activity and RhoA-dependent actomyosin contractility. Exp. Cell Res. 312, 387–400 (2006).
Wennerberg, K. & Der, C.J. Rho-family GTPases: it's not only Rac and Rho (and I like it). J. Cell Sci. 117, 1301–1312 (2004).
Krauthammer, M. et al. Exome sequencing identifies recurrent somatic RAC1 mutations in melanoma. Nat. Genet. 44, 1006–1014 (2012).
Kawazu, M. et al. Transforming mutations of RAC guanosine triphosphatases in human cancers. Proc. Natl. Acad. Sci. USA 110, 3029–3034 (2013).
Mitchison, T.J. & Cramer, L.P. Actin-based cell motility and cell locomotion. Cell 84, 371–379 (1996).
Lauffenburger, D.A. & Horwitz, A.F. Cell migration: a physically integrated molecular process. Cell 84, 359–369 (1996).
Olson, M.F., Paterson, H.F. & Marshall, C.J. Signals from Ras and Rho GTPases interact to regulate expression of p21Waf1/Cip1. Nature 394, 295–299 (1998).
Karlsson, R., Pedersen, E.D., Wang, Z. & Brakebusch, C. Rho GTPase function in tumorigenesis. Biochim. Biophys. Acta 1796, 91–98 (2009).
Gidekel Friedlander, S.Y. et al. Context-dependent transformation of adult pancreatic cells by oncogenic K-Ras. Cancer Cell 16, 379–389 (2009).
Borrman, R. in Handbuch der Speziellen Pathologische Anatomie und Histologie 4th edn. (ed. Henke, F. & Lubarsch, O.) 812–1054 (Verlag von Julius Springer, Berlin, 1926).
Saito, A., Shimoda, T., Nakanishi, Y., Ochiai, A. & Toda, G. Histologic heterogeneity and mucin phenotypic expression in early gastric cancer. Pathol. Int. 51, 165–171 (2001).
Henson, D.E., Dittus, C., Younes, M., Nguyen, H. & Albores-Saavedra, J. Differential trends in the intestinal and diffuse types of gastric carcinoma in the United States, 1973–2000: increase in the signet ring cell type. Arch. Pathol. Lab. Med. 128, 765–770 (2004).
Hayashi, T. et al. The superiority of the seventh edition of the TNM classification depends on the overall survival of the patient cohort: comparative analysis of the sixth and seventh TNM editions in patients with gastric cancer from Japan and the United Kingdom. Cancer 119, 1330–1337 (2013).
Kaneko, S. & Yoshimura, T. Time trend analysis of gastric cancer incidence in Japan by histological types, 1975–1989. Br. J. Cancer 84, 400–405 (2001).
Shang, X. et al. Rational design of small molecule inhibitors targeting RhoA subfamily Rho GTPases. Chem. Biol. 19, 699–710 (2012).
Shang, X. et al. Small-molecule inhibitors targeting G-protein-coupled Rho guanine nucleotide exchange factors. Proc. Natl. Acad. Sci. USA 110, 3155–3160 (2013).
Li, H. & Durbin, R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 25, 1754–1760 (2009).
Homer, N. & Nelson, S.F. Improved variant discovery through local re-alignment of short-read next-generation sequencing data using SRMA. Genome Biol. 11, R99 (2010).
Song, S. et al. qpure: A tool to estimate tumor cellularity from genome-wide single-nucleotide polymorphism profiles. PLoS ONE 7, e45835 (2012).
Carter, S.L. et al. Absolute quantification of somatic DNA alterations in human cancer. Nat. Biotechnol. 30, 413–421 (2012).
Wang, K., Li, M. & Hakonarson, H. ANNOVAR: functional annotation of genetic variants from high-throughput sequencing data. Nucleic Acids Res. 38, e164 (2010).
Qin, J., Jones, R.C. & Ramakrishnan, R. Studying copy number variations using a nanofluidic platform. Nucleic Acids Res. 36, e116 (2008).
Dube, S., Qin, J. & Ramakrishnan, R. Mathematical analysis of copy number variation in a DNA sample using digital PCR on a nanofluidic device. PLoS ONE 3, e2876 (2008).
Acknowledgements
We would like to thank K. Shiina, K. Sakuma, K. Nakano, A. Nishimoto and R. Sato for technical assistance. This study was supported by Grants-in-Aid for Scientific Research in Innovative Areas to the “Systems Cancer Project” (grant 22134003) from the Ministry of Education, Culture, Sports, Science and Technology of Japan (S.I.), by JSPS (Japan Society for the Promotion of Science) KAKENHI grant 24221011 (H.A.), by a Health Labor Sciences Research Grant (H.A.) and by a grant for the Translational Systems Biology and Medicine Initiative (TSBMI) from the Ministry of Education, Culture, Sports, Science and Technology of Japan (S.Y., K. Tatsuno and H.A.). The supercomputing resource was provided by the Human Genome Center (University of Tokyo).
Author information
Authors and Affiliations
Contributions
S.I. and H.A. designed the study. M.K. processed samples and supervised exome sequencing. H.U., K.G., K. Tatsuno and S.Y. performed computational analyses. Y.S., M.F., I.W., N.S., A.H. and S.N. coordinated sample acquisition. H.K., T.I. and T.U. carried out pathological review and analysis. A.T. performed dissection analysis. T.N. and S.F. performed functional experiments. Y.W. carried out structural analysis. S.I. wrote the manuscript. H.A., M.F., K. Tateishi and K.K. were involved in critical review and discussion.
Corresponding author
Ethics declarations
Competing interests
T.N. and S.F. are employees of Forerunner Pharma Research Co., Ltd. Y.W. is an employee of Chugai Pharmaceutical Co., Ltd.
Integrated supplementary information
Supplementary Figure 1 Tumor sample profiles for our exome sequencing analysis for 30 DGCs.
(a) Number of somatic mutations/indels and estimated tumor content ratio in each sample. N/A, less than 0.2 and could not be reliably calculated (see Online Method for details). (b) Number of somatic mutations/indels previously reported by Wang et al.9. MSI, microsatellite instable; MSS, microsatellite stable.
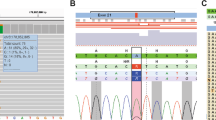
Supplementary Figure 2 Validation of discovered mutant alleles of variable frequency by other technologies.
(a) Concordance of RHOA mutant allele frequencies between Illumina exome, amplicon deep sequencing and digital PCR technologies. Mutations were detected in the discovery set by exome sequencing. Digital PCR was performed to validate RHOA mutations affecting the hotspots Y42 and R5. In the non-tumor sample GC_332, very low frequency for the mutated allele was observed by exome sequencing and TSCA. This may be due to a small number of tumor cells infiltrating into an apparently normal tissue area, as is often seen in DGC. ND, not detected. (b) Representative Sanger sequencing of samples with high and low mutant allele frequency. All of the cases with mutant allele frequency of >0.06 in TSCA data were also validated by Sanger sequencing. (c) Scatter plot of RHOA mutant allele frequencies from exome sequencing (x axis) and targeted amplicon deep sequencing (y axis). On this plot, there are seven mutants detected using defined criteria by both exome sequencing and TSCA and three mutants identified by TSCA only because of low allele frequency or low read depth at the positions in exome sequencing. (d) Sanger sequencing and targeted amplicon deep sequencing to validate the somatic nature of the mutations in all of the cases with G17V and R5Q, which are also present in the dbSNP database (rs11552761 and rs11552758).
Supplementary Figure 3 Mutational signatures of DGC.
(a) Mutational signature of each DGC sample in our exome sequencing. (b) Mutational signatures of IGCs in the previous report9. MSI and MSS are displayed separately. In both panels, each signature is displayed according to the 96-substitution classification defined by substitution patterns and sequence context. The probability bars for six types of substitutions are displayed in different colors. Samples were included in this analysis if more than 25 substitutions were detected among all in-target silent and non-silent somatic mutations that passed the quality filters. Red arrows represent GTG>GGG substitutions, and blue arrows represent CTT>CGT substitutions. *, pfg011T was classified as intestinal type according to Lauren's classification, but was histologically a poorly differentiated adenocarcinoma.
Supplementary Figure 4 Deregulated pathways in DGS and IGC suggested from this study.
Mutation frequencies in DGC and IGC are shown in red and black, respectively. RHOA and CDH1, which are specifically mutated in DGC, are shown with red circles. NM, nuclear membrane; PM, plasma membrane.
Supplementary Figure 5 Representative histology of DGSs with RHOA mutation.
(a–c) Histologic pictures of a representative case of DGC with RHOA mutation (GC_315). A low-power view (a) is shown together with higher-magnification views of the mucosal tubular component (b, upper square frame in a) and the deeper invasive poorly cohesive component (c, lower square frame in a). (d–g) Microscope-based dissection procedure. The intramucosal area (above the dotted line in d) and the deeper invasive area (below the dotted line in f) were dissected and analyzed for RHOA mutation separately. Slides were stained with hematoxylin and eosin after dissection of the mucosal area (e) and the deeper invasive area (g).
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5, Supplementary Tables 1–4 and 6, and Supplementary Note (PDF 1916 kb)
Supplementary Table 5
Summary statistics and data quality metrics for whole-exome sequencing and TruSeq Custom Amplicon sequencing for our discovery and validation cohort. (XLSX 59 kb)
Supplementary Data Set 1
Detailed somatic mutation data. (XLSX 549 kb)
Source data
Rights and permissions
About this article
Cite this article
Kakiuchi, M., Nishizawa, T., Ueda, H. et al. Recurrent gain-of-function mutations of RHOA in diffuse-type gastric carcinoma. Nat Genet 46, 583–587 (2014). https://doi.org/10.1038/ng.2984
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.2984
This article is cited by
-
Enhanced BCAT1 activity and BCAA metabolism promotes RhoC activity in cancer progression
Nature Metabolism (2023)
-
Predictive biomarkers in gastric cancer
Journal of Cancer Research and Clinical Oncology (2023)
-
Molecular profile of poorly cohesive gastric carcinoma with special reference to survival
Gastric Cancer (2023)
-
Multiancestry genomic and transcriptomic analysis of gastric cancer
Nature Genetics (2023)
-
RHOA protein expression correlates with clinical features in gastric cancer: a systematic review and meta-analysis
BMC Cancer (2022)