Abstract
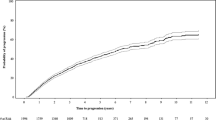
A markedly elevated serum free light chain (FLC) ratio may serve as a biomarker for malignant transformation in high-risk smoldering multiple myeloma (SMM) and identify patients who are at imminent risk of progression. We retrospectively studied the predictive value of the serum (FLC) assay in 586 patients with SMM diagnosed between 1970 to 2010. A serum involved/uninvolved FLC ratio ⩾100 was used to define high-risk SMM, which included 15% (n=90) of the total cohort. Receiver operating characteristics analysis determined the optimal FLC ratio cut-point to predict progression to symptomatic multiple myeloma (MM) within 2 years of diagnosis, which resulted in a specificity of 97% and sensitivity of 16%. Fifty-six percent of patients developed progressive disease during median follow-up of 52 months, but this increased to 98% in the subgroup of patients with FLC ratio ⩾100. The median time to progression in the FLC ratio ⩾100 group was 15 months versus 55 months in the FLC <100 group (P<0.0001). The risk of progression to MM within the first 2 years in patients with an FLC ratio ⩾100 was 72%; the risk of progression to MM or light chain amyloidosis in 2 years was 79%. We conclude that a high FLC ratio ⩾100 is a predictor of imminent progression in SMM, and such patients may be considered candidates for early treatment intervention.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Kyle RA, Durie BG, Rajkumar SV, Landgren O, Blade J, Merlini G et al. Monoclonal gammopathy of undetermined significance (MGUS) and smoldering (asymptomatic) multiple myeloma: IMWG consensus perspectives risk factors for progression and guidelines for monitoring and management. Leukemia 2010; 24: 1121–1127.
Group TIMW. Criteria for the classification of monoclonal gammopathies, multiple myeloma and related disorders: a report of the International Myeloma Working Group. Br J Haematol 2003; 121: 749–757.
Rajkumar SV, Gertz MA, Lacy MQ, Dispenzieri A, Fonseca R, Geyer SM et al. Thalidomide as initial therapy for early-stage myeloma. Leukemia 2003; 17: 775–779.
Barlogie B, van Rhee F, Shaughnessy JD, Epstein J, Yaccoby S, Pineda-Roman M et al. Seven-year median time to progression with thalidomide for smoldering myeloma: partial response identifies subset requiring earlier salvage therapy for symptomatic disease. Blood 2008; 112: 3122–3125.
Musto P, Petrucci MT, Bringhen S, Guglielmelli T, Caravita T, Bongarzoni V et al. A multicenter, randomized clinical trial comparing zoledronic acid versus observation in patients with asymptomatic myeloma. Cancer 2008; 113: 1588–1595.
Rajkumar SV, Merlini G, San Miguel JF . Redefining myeloma. Nat Rev Clin Oncol 2012; 9: 494–496.
Kyle RA, Remstein ED, Therneau TM, Dispenzieri A, Kurtin PJ, Hodnefield JM et al. Clinical course and prognosis of smoldering (asymptomatic) multiple myeloma. N Engl J Med 2007; 356: 2582–2590.
Kyle RA, Therneau TM, Rajkumar SV, Offord JR, Larson DR, Plevak MF et al. A long-term study of prognosis in monoclonal gammopathy of undetermined significance. N Engl J Med 2002; 346: 564–569.
Perez-Persona E, Vidriales MB, Mateo G, Garcia-Sanz R, Mateos MV, de Coca AG et al. New criteria to identify risk of progression in monoclonal gammopathy of uncertain significance and smoldering multiple myeloma based on multiparameter flow cytometry analysis of bone marrow plasma cells. Blood 2007; 110: 2586–2592.
Rajkumar SV, Larson D, Kyle RA . Diagnosis of smoldering multiple myeloma. N Engl J Med 2011; 365: 474–475.
Dispenzieri A, Kyle RA, Katzmann JA, Therneau TM, Larson D, Benson J et al. Immunoglobulin free light chain ratio is an independent risk factor for progression of smoldering (asymptomatic) multiple myeloma. Blood 2008; 111: 785–789.
Rajkumar SV, Kyle RA, Therneau TM, Clark RJ, Bradwell AR, Melton LJ et al. Presence of monoclonal free light chains in the serum predicts risk of progression in monoclonal gammopathy of undetermined significance. Br J Haematol 2004; 127: 308–310.
Snozek CL, Katzmann JA, Kyle RA, Dispenzieri A, Larson DR, Therneau TM et al. Prognostic value of the serum free light chain ratio in newly diagnosed myeloma: proposed incorporation into the international staging system. Leukemia 2008; 22: 1933–1937.
Lachmann HJ, Gallimore R, Gillmore JD, Carr-Smith HD, Bradwell AR, Pepys MB et al. Outcome in systemic AL amyloidosis in relation to changes in concentration of circulating free immunoglobulin light chains following chemotherapy. Br J Haematol 2003; 122: 78–84.
Katzmann JA, Abraham RS, Dispenzieri A, Lust JA, Kyle RA . Diagnostic performance of quantitative kappa and lambda free light chain assays in clinical practice. Clin Chem 2005; 51: 878–881.
Katzmann JA, Clark RJ, Abraham RS, Bryant S, Lymp JF, Bradwell AR et al. Serum reference intervals and diagnostic ranges for free kappa and free lambda immunoglobulin light chains: relative sensitivity for detection of monoclonal light chains. Clin Chem 2002; 48: 1437–1444.
Bradwell AR, Carr-Smith HD, Mead GP, Harvey TC, Drayson MT . Serum test for assessment of patients with Bence Jones myeloma. Lancet 2003; 361: 489–491.
Drayson M, Tang LX, Drew R, Mead GP, Carr-Smith H, Bradwell AR . Serum free light-chain measurements for identifying and monitoring patients with nonsecretory multiple myeloma. Blood 2001; 97: 2900–2902.
van Rhee F, Bolejack V, Hollmig K, Pineda-Roman M, Anaissie E, Epstein J et al. High serum-free light chain levels and their rapid reduction in response to therapy define an aggressive multiple myeloma subtype with poor prognosis. Blood 2007; 110: 827–832.
Waxman AJ, Kuehl M, Balakumaran A, Weiss B, Landgren O . Smoldering (asymptomatic) multiple myeloma: revisiting the clinical dilemma and looking into the future. Clin Lymphoma Myeloma Leuk 2010; 10: 248–257.
Alexanian R, Barlogie B, Dixon D . Prognosis of asymptomatic multiple myeloma. Arch Intern Med 1988; 148: 1963–1965.
Lust JA, Lacy MQ, Zeldenrust SR, Dispenzieri A, Gertz MA, Witzig TE et al. Induction of a chronic disease state in patients with smoldering or indolent multiple myeloma by targeting interleukin 1{beta}-induced interleukin 6 production and the myeloma proliferative component. Mayo Clin Proc 2009; 84: 114–122.
Golombick T, Diamond TH, Manoharan A, Ramakrishna R . Monoclonal gammopathy of undetermined significance, smoldering multiple myeloma, and curcumin: a randomized, double-blind placebo-controlled cross-over 4 g study and an open-label 8 g extension study. Am J Hematol 2012; 87: 455–460.
Landgren O, Kyle RA, Pfeiffer RM, Katzmann JA, Caporaso NE, Hayes RB et al. Monoclonal gammopathy of undetermined significance (MGUS) consistently precedes multiple myeloma: a prospective study. Blood 2009; 113: 5412–5417.
Mateos M, Lopez-Corral L, Hernandez M, Giraldo P, De la Rubia J, De Arriba F et al. Smoldering multiple myeloma (SMM) at high-risk of progression to symptomatic disease: a phase III, randomized, multicenter trial based on lenalidomide-dexamethasone (Len-Dex) as induction therapy followed by maintenance therapy with Len alone vs no treatment. Blood (ASH Annual Meeting Abstracts) 2011; 118: 3996.
Acknowledgements
This work was supported in part by the Jabbs Foundation (Birmingham, UK); National Cancer Institute grants CA168762, CA 107476, CA 62242, CA 100707, CA 83724, and the Henry J Predolin Foundation, USA.
Author contributions
JTL, SKK and SVR designed the research, analyzed the data, wrote and edited the manuscript. RAK, JAK and AD participated in data interpretation, reviewed the manuscript and provided critical comments. All authors reviewed and approved the final manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Larsen, J., Kumar, S., Dispenzieri, A. et al. Serum free light chain ratio as a biomarker for high-risk smoldering multiple myeloma. Leukemia 27, 941–946 (2013). https://doi.org/10.1038/leu.2012.296
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2012.296
Keywords
This article is cited by
-
Smoldering multiple myeloma current treatment algorithms
Blood Cancer Journal (2022)
-
Monoclonal proteinuria predicts progression risk in asymptomatic multiple myeloma with a free light chain ratio ≥100
Leukemia (2022)
-
Heterogeneity in the diagnosis of plasmablastic lymphoma, plasmablastic myeloma, and plasmablastic neoplasm: a scoping review
International Journal of Hematology (2021)
-
The molecular make up of smoldering myeloma highlights the evolutionary pathways leading to multiple myeloma
Nature Communications (2021)
-
Comparison of three different serum-free light-chain assays—implications on diagnostic and therapeutic monitoring of multiple myeloma
Blood Cancer Journal (2020)