Abstract
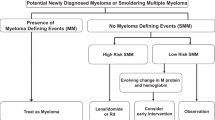
Smoldering multiple myeloma (SMM) is an asymptomatic plasma cell disorder characterized by the presence of one or both features of serum M-protein ≥ 30 g/L and bone marrow plasma cell infiltration ≥ 10 %. However, myeloma-related symptomatology is absent from this condition. The risk of progression to active MM is not uniform, and several markers are useful for identifying SMM patients at high risk of progression to active MM. These include the size of the M-protein and the infiltration in the bone marrow, the serum-free light-chain ratio, the presence of immunoparesis and percentage of plasma cell with aberrant phenotype by flow cytometry, or the presence of focal lesions in magnetic resonance imaging. Overall, the presence of these factors identifies patients who have a 50 % probability of progression at 2 years, and the forthcoming challenge will be to identify ultra-high-risk patients who have an 80 % risk of progression at 2 years. The current standard of care is not to treat until progression to symptomatic disease occurs. Several trials of melphalan, thalidomide and bisphosphonates have been conducted in the overall SMM patient population to examine the delay in time to progression (TTP) to symptomatic disease, but these have shown no significant benefit. However, a randomized trial that focused on high-risk SMM patients allocated to receive early treatment with lenalidomide plus dexamethasone versus observation did report a significant benefit with respect to TTP and overall survival. In summary, high-risk SMM patients should be targetted for early treatment, and more so efforts should be made to identify the ultra-high-risk subgroup within the high-risk SMM patient population which may be considered as early MM and thereby candidates for receiving therapy before they develop myeloma-related symptomatology.
Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
Kyle RA, Greipp PR. Smoldering multiple myeloma. N Engl J Med. 1980;302(24):1347–9.
Criteria for the classification of monoclonal gammopathies, multiple myeloma and related disorders: a report of the International Myeloma Working Group. Br J Haematol. 2003;121(5):749–57.
• Kyle RA, Rajkumar SV. Criteria for diagnosis, staging, risk stratification and response assessment of multiple myeloma. Leukemia. 2009;23(1):3–9. The first and largest systematic study focusing on SMM that identified different risk subgroups.
Blade J, Dimopoulos M, Rosinol L, Rajkumar SV, Kyle RA. Smoldering (asymptomatic) multiple myeloma: current diagnostic criteria, new predictors of outcome, and follow-up recommendations. J Clin Oncol. 2010;28(4):690–7.
Kyle RA, Remstein ED, Therneau TM, Dispenzieri A, Kurtin PJ, Hodnefield JM, et al. Clinical course and prognosis of smoldering (asymptomatic) multiple myeloma. N Engl J Med. 2007;356(25):2582–90.
• Perez-Persona E, Vidriales MB, Mateo G, Garcia-Sanz R, Mateos MV, de Coca AG, et al. New criteria to identify risk of progression in monoclonal gammopathy of uncertain significance and smoldering multiple myeloma based on multiparameter flow cytometry analysis of bone marrow plasma cells. Blood. 2007;110(7):2586–92. Definition of new criteria to evaluate the risk of progression from asymptomatic to symptomatic multiple myeloma.
Perez-Persona E, Mateo G, Garcia-Sanz R, Mateos MV, de Las Heras N, de Coca AG, et al. Risk of progression in smouldering myeloma and monoclonal gammopathies of unknown significance: comparative analysis of the evolution of monoclonal component and multiparameter flow cytometry of bone marrow plasma cells. Br J Haematol. 2010;148(1):110–4.
Dispenzieri A, Kyle RA, Katzmann JA, Therneau TM, Larson D, Benson J, et al. Immunoglobulin free light chain ratio is an independent risk factor for progression of smoldering (asymptomatic) multiple myeloma. Blood. 2008;111(2):785–9.
Larsen JT, Kumar SK, Dispenzieri A, Kyle RA, Katzmann JA, Rajkumar SV. Serum free light chain ratio as a biomarker for high-risk smoldering multiple myeloma. Leukemia. 2013;27(4):941–6.
Bianchi G, Kyle RA, Larson DR, Witzig TE, Kumar S, Dispenzieri A, et al. High levels of peripheral blood circulating plasma cells as a specific risk factor for progression of smoldering multiple myeloma. Leukemia. 2013;27(3):680–5.
Rajkumar SV, Larson D, Kyle RA. Diagnosis of smoldering multiple myeloma. N Engl J Med. 2011;365(5):474–5.
Rosinol L, Blade J, Esteve J, Aymerich M, Rozman M, Montoto S, et al. Smoldering multiple myeloma: natural history and recognition of an evolving type. Br J Haematol. 2003;123(4):631–6.
Moulopoulos LA, Dimopoulos MA, Smith TL, Weber DM, Delasalle KB, Libshitz HI, et al. Prognostic significance of magnetic resonance imaging in patients with asymptomatic multiple myeloma. J Clin Oncol. 1995;13(1):251–6.
Hillengass J, Kilk K, Listl K, Hielscher T, Neben K, Hose D, et al. Over 30% of Smoldering Myeloma Patients Have Tumor Cell Bone Marrow Infiltration Patterns Similar to Multiple Myeloma: A Large (n=544) Clinical Study Using Whole-Body MRI. ASH Annual Meeting Abstracts. 2012;120(21):2911.
Hillengass J, Fechtner K, Weber MA, Bauerle T, Ayyaz S, Heiss C, et al. Prognostic significance of focal lesions in whole-body magnetic resonance imaging in patients with asymptomatic multiple myeloma. J Clin Oncol. 2010;28(9):1606–10.
Neben K, Jauch A, Hielscher T, Hillengass J, Lehners N, Raab MS, et al. The Chromosomal Abnormalities Del(17p), t(4;14), and +1q21 Predict Progression From Smoldering to Symptomatic Multiple Myeloma. ASH Annual Meeting Abstracts. 2012;120(21):1806.
Rajkumar SV, Gupta V, Fonseca R, Dispenzieri A, Gonsalves WI. Larson D, et al. Leukemia: Impact of primary molecular cytogenetic abnormalities and risk of progression in smoldering multiple myeloma; 2013.
Hjorth M, Hellquist L, Holmberg E, Magnusson B, Rodjer S, Westin J. Initial versus deferred melphalan-prednisone therapy for asymptomatic multiple myeloma stage I–a randomized study. Myeloma Group of Western Sweden. Eur J Haematol. 1993;50(2):95–102.
Grignani G, Gobbi PG, Formisano R, Pieresca C, Ucci G, Brugnatelli S, et al. A prognostic index for multiple myeloma. Br J Cancer. 1996;73(9):1101–7.
Riccardi A, Mora O, Tinelli C, Valentini D, Brugnatelli S, Spanedda R, et al. Long-term survival of stage I multiple myeloma given chemotherapy just after diagnosis or at progression of the disease: a multicentre randomized study. Cooperative Group of Study and Treatment of Multiple Myeloma. Br J Cancer. 2000;82(7):1254–60.
Rajkumar SV, Gertz MA, Lacy MQ, Dispenzieri A, Fonseca R, Geyer SM, et al. Thalidomide as initial therapy for early-stage myeloma. Leukemia. 2003;17(4):775–9.
Weber D, Rankin K, Gavino M, Delasalle K, Alexanian R. Thalidomide alone or with dexamethasone for previously untreated multiple myeloma. J Clin Oncol. 2003;21(1):16–9.
Barlogie B, van Rhee F, Shaughnessy Jr JD, Epstein J, Yaccoby S, Pineda-Roman M, et al. Seven-year median time to progression with thalidomide for smoldering myeloma: partial response identifies subset requiring earlier salvage therapy for symptomatic disease. Blood. 2008;112(8):3122–5.
Witzig TE, Laumann KM, Lacy MQ, Hayman SR, Dispenzieri A, Kumar S, et al. A phase III randomized trial of thalidomide plus zoledronic acid versus zoledronic acid alone in patients with asymptomatic multiple myeloma. Leukemia. 2013;27(1):220–5.
Martin A, Garcia-Sanz R, Hernandez J, Blade J, Suquia B, Fernandez-Calvo J, et al. Pamidronate induces bone formation in patients with smouldering or indolent myeloma, with no significant anti-tumour effect. BrJHaematol. 2002;118(1):239–42.
D'Arena G, Gobbi PG, Broglia C, Sacchi S, Quarta G, Baldini L, et al. Pamidronate versus observation in asymptomatic myeloma: final results with long-term follow-up of a randomized study. Leuk Lymphoma. 2011;52(5):771–5.
Musto P, Petrucci MT, Bringhen S, Guglielmelli T, Caravita T, Bongarzoni V, et al. A multicenter, randomized clinical trial comparing zoledronic acid versus observation in patients with asymptomatic myeloma. Cancer. 2008;113(7):1588–95.
Mateos MV, Hernández MT, Giraldo P, de La Rubia J, de Arriba F, López-Corral L, et al. Lenalidomide plus Dexamethasone for High-Risk Smoldering Multiple Myeloma. N Engl J Med. 2013;369:438–47.
• Cherry BM, Korde N, Kwok M, Manasanch EE, Bhutani M, Mulquin M, et al. Modeling progression risk for smoldering multiple myeloma: results from a prospective clinical study. Leuk Lymphoma. 2013 Jan 11. The relevance of this paper is to note that there is necessary to built homogeneous modeling progression risk for smoldering myeloma including clinical and biological markers.
Compliance with Ethics Guidelines
Conflict of Interest
M. Mateos: received payment by lectures for Celgene Corporation. J. San Miguel: participated in advisory boards and lectures for Celgene, Janssen, Novartis, Millennium.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mateos, MV., San Miguel, J.F. New Approaches to Smoldering Myeloma. Curr Hematol Malig Rep 8, 270–276 (2013). https://doi.org/10.1007/s11899-013-0174-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11899-013-0174-1