Abstract
Purpose
To prospectively assess whether choline levels and Apparent Diffusion Coefficient (ADC) values within cervical cancers before, during, and after non-surgical therapy are predictive of tumour response.
Patients and methods
Patients undergoing MR examination for staging of cervical cancer, candidate for non-surgical therapy, were prospectively enrolled. According to the status at the end of therapies, patients were divided into responders and non-responders. The final outcome after a 5-year follow-up was classified as No Evidence of Disease (NED) or Progression of Disease (PD). Baseline values of mean ADC and Cho/H2O were compared between responders and non-responders, as well as between patients with NED and PD. The percent variation of ADC and Cho/H2O values over time was compared. P values <0.05 were considered significant.
Results
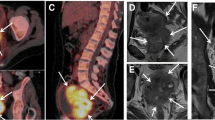
16 patients were included. There was no significant difference at baseline between responders (n = 12) and non-responders (n = 4), nor between NED (n = 11) PD patients (n = 5), in ADC values and Cho/H2O ratio. There was no significant difference in percent variation of ADC values and of Cho/H2O, comparing responders and non-responders. There was a significant increase in absolute values of ADC from the initial to mid-therapy MRI (p = 0.0001), while Cho/H2O was stable (p value: 0.61). In the four non-responders, the ADC increase was not significant (p value: 0.25), while it was significant in the 11 responders (p value: 0.001). Values of spectroscopy were stable in both responders and non-responders.
Conclusions
High increases of ADC values from baseline to mid-therapy MR reflect response to therapies. There were no significant variations in choline/water ratios over time.


Similar content being viewed by others
References
Siegel RL, Miller KD, Jemal A (2015) CA Cancer J Clin. Jan-Feb;65(1):5-29. doi:10.3322/caac.21254 Cancer statistics, 2015
Klostergaard J, Parga K, Raptis RG (2010) Current and future applications of magnetic resonance imaging (MRI) to breast and ovarian cancer patient management. PR Health Sci J 29(3):223–231
Koh DM, Collins DJ (2007) Diffusion-weighted MRI in the body: applications and challenges in oncology. AJR Am J Roentgenol 188(6):1622–1635
Heo SH, Shin SS, Kim JW, Lim HS, Jeong YY, Kang WD et al (2013) Pre-treatment diffusion-weighted MR imaging for predicting tumor recurrence in uterine cervical cancer treated with concurrent chemoradiation: value of histogram analysis of apparent diffusion coefficients. Korean J Radiol 14(4):616–625
Shah N, Sattar A, Benanti M, Hollander S, Cheuck L (2006) Magnetic resonance spectroscopy as an imaging tool for cancer: a review of the literature. J Am Osteopath Assoc 106:23–27
Pinker K, Stadlbauer A, Bogner W, Gruber S, Helbich TH (2012) Molecular imaging of cancer: MR spectroscopy and beyond. Eur J Radiol Mar;81(3):566–577
Payne GS, Schmidt M, Morgan VA, Giles S, Bridges J, Ind T, DeSouza NM (2010) Evaluation of magnetic resonance diffusion and spectroscopy measurements as predictive biomarkers in stage 1 cervical cancer. Gynecol Oncol Feb;116(2):246–52
Schwarz AJ, Maisey NR, Collins DJ, Cunningham D, Huddart R, Leach MO (2002) Early in vivo detection of metabolic response: a pilot study of 1H MR spectroscopy in extracranial lymphoma and germ cell tumours. Br J Radiol Dec;75(900): 959–66
Haddadin IS, McIntosh A, Meisamy S, Corum C, Styczynski Snyder AL, Powell NJ, et al (2009) Metabolite quantification and high-field MRS in breast cancer. NMR Biomed 22(1):65–76
Meisamy S, Bolan PJ, Baker EH et al (2004) Neoadjuvant chemotherapy of locally advanced breast cancer: predicting response with in vivo (1)H MR spectroscopy—a pilot study at 4 T. Radiology 233(2):424–431
Pecorelli S, Zigliani L, Odicino F (2009) Revised FIGO staging for carcinoma of the cervix. Int J Gynaecol Obstet 105:107–108
Zhao M, Pipe JG, Bonnett J, Evelhoch JL (1996) Early detection of treatment response by diffusion-weighted 1H-NMR spectroscopy in a murine tumour in vivo. Br J Cancer 73:61–64
Huang MQ, Nelson DS, Pickup S, Qiao H, Delikatny EJ, Poptani H et al (2007) In vivo monitoring response to chemotherapy of human diffuse large B-cell lymphoma xenografts in SCID mice by 1H and 31P MRS. Acad Radiol 14:1531–1539
Morse DL, Raghunand N, Sadarangani P, Murthi S, Job C, Day S et al (2007) Response of choline metabolites to docetaxel therapy is quantified in vivo by localized (31)P MRS of human breast cancer xenografts and in vitro by high-resolution (31)P NMR spectroscopy of cell extracts. Magn Reson Med 58:270–280
Manton DJ, Chaturvedi A, Hubbard A, Lind MJ, Lowry M, Maraveyas A et al (2006) Neoadjuvant chemotherapy in breast cancer: early response prediction with quantitative MR imaging and spectroscopy. Br J Cancer 94:427–435
Wu B, Peng WJ, Wang PJ, Gu YJ, Li WT, Zhou LP et al (2006) In vivo 1H magnetic resonance spectroscopy in evaluation of hepatocellular carcinoma and its early response to transcatheter arterial chemoembolization. Chin Med Sci J 21:258–264
Preul MC, Caramanos Z, Villemure JG, Shenouda G, LeBlanc R, Langleben A et al (2000) Using proton magnetic resonance spectroscopic imaging to predict in vivo the response of recurrent malignant gliomas to tamoxifen chemotherapy. Neurosurgery 46:306–318
Booth SJ, Pickles MD, Turnbull LW (2009) In vivo magnetic resonance spectroscopy of gynaecological tumours at 3.0 Tesla. BJOG 116(2):300–303
Mahon MM, Cox IJ, Dina R et al (2004) (1) H magnetic resonance spectroscopy of preinvasive and invasive cervical cancer: in vivo-ex vivo profiles and effect of tumor load. J Magn Reson Imaging 19(3):356–364
deSouza NM, McIndoe GA, Soutter WP, Krausz T, Chui KM, Hughes C et al (1998) Value of magnetic resonance imaging with an endovaginal receiver coil in the preoperative assessment of Stage I and IIa cervical neoplasia. Br J Obstet Gynaecol 105(5):500–507
Allen JR, Prost RW, Griffith OW, Erickson SJ, Erickson BA (2001) In vivo proton (H1) magnetic resonance spectroscopy for cervical carcinoma. Am J Clin Oncol 24(5):522–529
Zhu M, Fischl AS, Trowbridge MA, Shannon HE (2012) Reproducibility of total choline/water ratios in mouse U87MG xenograft tumors by 1H-MRS. J Magn Reson Imaging 36(2):459–467
Hamstra DA, Rehemtulla A, Ross BD (2007) Diffusion magnetic resonance imaging: abiomarker for treatment response in oncology. J Clin Oncol 25:4104–4109
Koh DM, Padhani AR (2006) Diffusion-weighted MRI. A new functional clinical technique for tumour imaging. Br J Radiol 79:633–635
Pickles MD, Gibbs P, Lowry M, Turnbull LW (2006) Diffusion changes precede sizereduction in neoadjuvant treatment of breast cancer. Magn Reson Imaging 24:843–847
Deng J, Miller FH, Rhee TK, Sato KT, Mulcahy MF, Kulik LM et al (2006) Diffusion weighted MR imaging for determination of hepatocellular carcinoma response toyttrium-90 radioembolization. J Vasc Interv Radiol 17:1195–1200
Naganawa S, Sato C, Kumada H, Ishigaki T, Miura S, Takizawa O (2005) Apparent diffusion coefficient in cervical cancer of the uterus: comparison with the normal uterine cervix. Eur Radiol 15:71–78
Rizzo S, Summers P, Raimondi S, Belmonte M, Maniglio M, Landoni F, Colombo N, Bellomi M (2011) Diffusion-weighted MR imaging in assessing cervical tumour response to nonsurgical therapy. Radiol Med 116(5):766–780
McVeigh PZ, Syed AM, Milosevic M, Fyles A, Haider MA (2008) Diffusion weighted MRI in cervical cancer. Eur Radiol 18:1058–1064
Harry VN, Semple SI, Gilbert FJ, Parkin DE (2008) Diffusion-weighted magnetic resonance imaging in the early detection of response to chemoradiation in cervical cancer. Gynecol Oncol 111(2):213–220
Das S, Chandramohan A (2015) Rami Reddy JK, Mukhopadhyay S, Kumar RM, Isiah R, John S, Oommen R, Jeyaseelan V. Role of conventional and diffusion weighted MRI in predicting treatment response after low dose radiation and chemotherapy in locally advanced carcinoma cervix. Radiother Oncol 117(2):288–293
SEER Stat Fact Sheets: Cervix Uteri Cancer (2016). http://seer.cancer.gov/statfacts/html/cervix.html. Accessed 12/05/2016
Osman M (2014) The role of neoadjuvant chemotherapy in the management of locally advanced cervix cancer: a systematic review.Oncol Rev. Sep 23;8(2):250. doi:10.4081/oncol.2014.250. eCollection 2014
Padhani AR, Liu G, Koh DM et al (2009) Diffusion-weighted magnetic resonance imaging as a cancer biomarker: consensus and recommendations. Neoplasia 11:102–125
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Statement of human rights
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Rights and permissions
About this article
Cite this article
Rizzo, S., Buscarino, V., Origgi, D. et al. Evaluation of diffusion-weighted imaging (DWI) and MR spectroscopy (MRS) as early response biomarkers in cervical cancer patients. Radiol med 121, 838–846 (2016). https://doi.org/10.1007/s11547-016-0665-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11547-016-0665-y