Abstract
Purpose
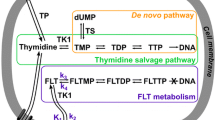
Positron emission tomography with the thymidine analogue 3′-deoxy-3′-[18F]fluorothymidine (FLT) has been reported to closely reflect lymphoma proliferation in vivo. In this preclinical study, we have investigated if FLT can also be utilized for imaging therapy-induced alterations of the nucleoside metabolism and if FLT is a surrogate marker for early response to cytotoxic treatment.
Materials and Methods
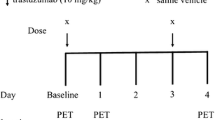
Immunodeficient mice bearing high-grade lymphoma xenotransplants were treated with the cytotoxic agent doxorubicin (day 0). In the time course of day +1 to +9, antiproliferative effects were assessed non-invasively with FLT-PET and correlated to changes of the proliferation fraction and induction of apoptosis, as assessed by immunohistochemistry.
Results
Tumor growth in untreated animals was significantly higher than in treated animals. In FLT-PET scans, these observations correlated with a significant decrease of tumor-to-background ratio in the therapy group already at day 1. Likewise, median tumor-to-muscle ratio of FLT uptake already declined at day 1. The proliferation fraction assessed by Ki-67 immunohistochemistry decreased after chemotherapy, while activated caspase 3 increased, suggesting both cell cycle arrest and induction of apoptosis as underlying mechanisms of the observed PET-signal alterations.
Conclusion
In a lymphoma xenotransplant model, we show that positron emission tomography using the proliferation marker FLT is suitable to detect early response to cytotoxic treatment. A significant decrease of FLT uptake but not tumor growth was detectable already 24 h after therapy and correlated with reduced proliferation and induction of apoptosis. Thus, FLT-PET has a potential for imaging early response to treatment in malignant lymphoma.





Similar content being viewed by others
References
Cheson BD, Horning SJ, Coiffier B et al (1999) Report of an international workshop to standardize response criteria for non-Hodgkin's lymphomas. NCI Sponsored International Working Group. J Clin Oncol 17:1244
Lister TA, Crowther D, Sutcliffe SB et al (1989) Report of a committee convened to discuss the evaluation and staging of patients with Hodgkin's disease: Cotswolds meeting. J Clin Oncol 7:1630–1636
Surbone A, Longo DL, DeVita VT Jr et al (1988) Residual abdominal masses in aggressive non-Hodgkin's lymphoma after combination chemotherapy: significance and management. J Clin Oncol 6:1832–1837
Jerusalem G, Beguin Y, Fassotte MF et al (1999) Whole-body positron emission tomography using 18F-fluorodeoxyglucose for posttreatment evaluation in Hodgkin's disease and non-Hodgkin's lymphoma has higher diagnostic and prognostic value than classical computed tomography scan imaging. Blood 94:429–433
Juweid ME, Stroobants S, Hoekstra OS et al (2007) Use of positron emission tomography for response assessment of lymphoma: consensus of the Imaging Subcommittee of International Harmonization Project in Lymphoma. J Clin Oncol 25:571–578
Kazama T, Faria SC, Varavithya V, Phongkitkarun S, Ito H, Macapinlac HA (2005) FDG PET in the evaluation of treatment for lymphoma: clinical usefulness and pitfalls. Radiographics 25:191–207
Sandherr M, von Schilling C, Link T et al (2001) Pitfalls in imaging Hodgkin's disease with computed tomography and positron emission tomography using fluorine-18-fluorodeoxyglucose. Ann Oncol 12:719–722
van Waarde A, Cobben DC, Suurmeijer AJ et al (2004) Selectivity of 18F-FLT and 18F-FDG for differentiating tumor from inflammation in a rodent model. J Nucl Med 45:695–700
Martiat P, Ferrant A, Labar D et al (1988) In vivo measurement of carbon-11 thymidine uptake in non-Hodgkin's lymphoma using positron emission tomography. J Nucl Med 29:1633–1637
Shields AF, Mankoff DA, Link JM et al (1998) Carbon-11-thymidine and FDG to measure therapy response. J Nucl Med 39:1757–1762
Shields AF, Grierson JR, Dohmen BM et al (1998) Imaging proliferation in vivo with [F-18]FLT and positron emission tomography. Nat Med 4:1334–1336
Barthel H, Perumal M, Latigo J et al (2005) The uptake of 3′-deoxy-3′-[18F]fluorothymidine into L5178Y tumours in vivo is dependent on thymidine kinase 1 protein levels. Eur J Nucl Med Mol Imaging 32:257–263
Kong XB, Zhu QY, Vidal PM et al (1992) Comparisons of anti-human immunodeficiency virus activities, cellular transport, and plasma and intracellular pharmacokinetics of 3′-fluoro-3′-deoxythymidine and 3′-azido-3′-deoxythymidine. Antimicrob Agents Chemother 36:808–818
Sherley JL, Kelly TJ (1988) Regulation of human thymidine kinase during the cell cycle. J Biol Chem 263:8350–8358
Eriksson S, Arner E, Spasokoukotskaja T et al (1994) Properties and levels of deoxynucleoside kinases in normal and tumor cells; implications for chemotherapy. Adv Enzyme Regul 34:13–25
Seitz U, Wagner M, Neumaier B et al (2002) Evaluation of pyrimidine metabolising enzymes and in vitro uptake of 3′-[(18)F]fluoro-3′-deoxythymidine ([(18)F]FLT) in pancreatic cancer cell lines. Eur J Nucl Med Mol Imaging 29:1174–1181
Rasey JS, Grierson JR, Wiens LW, Kolb PD, Schwartz JL (2002) Validation of FLT uptake as a measure of thymidine kinase-1 activity in A549 carcinoma cells. J Nucl Med 43:1210–1217
Lu L, Samuelsson L, Bergstrom M, Sato K, Fasth KJ, Langstrom B (2002) Rat studies comparing 11C-FMAU, 18F-FLT, and 76Br-BFU as proliferation markers. J Nucl Med 43:1688–1698
Chen W, Cloughesy T, Kamdar N et al (2005) Imaging proliferation in brain tumors with 18F-FLT PET: comparison with 18F-FDG. J Nucl Med 46:945–952
Cobben DC, Elsinga PH, Suurmeijer AJ et al (2004) Detection and grading of soft tissue sarcomas of the extremities with (18)F-3′-fluoro-3′-deoxy-L-thymidine. Clin Cancer Res 10:1685–1690
Smyczek-Gargya B, Fersis N, Dittmann H et al (2004) PET with [18F]fluorothymidine for imaging of primary breast cancer: a pilot study. Eur J Nucl Med Mol Imaging 31:720–724
Wagner M, Seitz U, Buck A et al (2003) 3′-[18F]fluoro-3′-deoxythymidine ([18F]-FLT) as positron emission tomography tracer for imaging proliferation in a murine B-cell lymphoma model and in the human disease. Cancer Res 63:2681–2687
Buck AK, Bommer M, Stilgenbauer S et al (2006) Molecular imaging of proliferation in malignant lymphoma. Cancer Res 66:11055–11061
Herrmann K, Wieder HA, Buck AK et al (2007) Early response assessment using 3′-deoxy-3′-[18F]fluorothymidine-positron emission tomography in high-grade non-Hodgkin′s lymphoma. Clin Cancer Res 13:3552–3558
Buck AK, Kratochwil C, Glatting G et al (2007) Early assessment of therapy response in malignant lymphoma with the thymidine analogue [(18)F]FLT. Eur J Nucl Med Mol Imaging 34:1775–1782
Momparler RL, Karon M, Siegel SE, Avila F (1976) Effect of adriamycin on DNA, RNA, and protein synthesis in cell-free systems and intact cells. Cancer Res 36:2891–2895
Di Bartolomeo S, Di Sano F, Piacentini M, Spinedi A (2000) Apoptosis induced by doxorubicin in neurotumor cells is divorced from drug effects on ceramide accumulation and may involve cell cycle-dependent caspase activation. J Neurochem 75:532–539
Zhou M, Gu L, Li F, Zhu Y, Woods WG, Findley HW (2002) DNA damage induces a novel p53-survivin signaling pathway regulating cell cycle and apoptosis in acute lymphoblastic leukemia cells. J Pharmacol Exp Ther 303:124–131
Kano Y, Akutsu M, Tsunoda S et al (2001) In vitro cytotoxic effects of a tyrosine kinase inhibitor STI571 in combination with commonly used antileukemic agents. Blood 97:1999–2007
Yang YJ, Ryu JS, Kim SY et al (2006) Use of 3′-deoxy-3′-[18F]fluorothymidine PET to monitor early responses to radiation therapy in murine SCCVII tumors. Eur J Nucl Med Mol Imaging 33:412–419
Apisarnthanarax S, Alauddin MM, Mourtada F et al (2006) Early detection of chemoradioresponse in esophageal carcinoma by 3′-deoxy-3′-3H-fluorothymidine using preclinical tumor models. Clin Cancer Res 12:4590–4597
Leyton J, Latigo JR, Perumal M, Dhaliwal H, He Q, Aboagye EO (2005) Early detection of tumor response to chemotherapy by 3′-deoxy-3′-[18F]fluorothymidine positron emission tomography: the effect of cisplatin on a fibrosarcoma tumor model in vivo. Cancer Res 65:4202–4210
Kenny L, Coombes RC, Vigushin DM, Al-Nahhas A, Shousha S, Aboagye EO (2007) Imaging early changes in proliferation at 1 week post chemotherapy: a pilot study in breast cancer patients with 3′-deoxy-3′-[(18)F]fluorothymidine positron emission tomography. Eur J Nucl Med Mol Imaging 34:1339–1347
Troost EG, Vogel WV, Merkx MA et al (2007) 18F-FLT PET does not discriminate between reactive and metastatic lymph nodes in primary head and neck cancer patients. J Nucl Med 48:726–735
Wieder HA, Geinitz H, Rosenberg R et al (2007) PET imaging with [(18)F]3′-deoxy-3′-fluorothymidine for prediction of response to neoadjuvant treatment in patients with rectal cancer. Eur J Nucl Med Mol Imaging 34:878–883
Kasper B, Egerer G, Gronkowski M et al (2007) Functional diagnosis of residual lymphomas after radiochemotherapy with positron emission tomography comparing FDG- and FLT-PET. Leuk lymphoma 48:746–753
Acknowledgements
We appreciate the excellent contributions made by our colleague Petra Watzlowik PhD and the great support by our technical staff members Sybille Reder, Elisabeth Aywanger, and Brigitte Dzewas. This work was supported by “EMIL” (European Molecular Imaging Laboratories, grant LSHC-CT-2004-503569).
Author information
Authors and Affiliations
Corresponding author
Additional information
Nicolas Graf and Ken Herrmann contributed equally to this work.
Rights and permissions
About this article
Cite this article
Graf, N., Herrmann, K., den Hollander, J. et al. Imaging Proliferation to Monitor Early Response of Lymphoma to Cytotoxic Treatment. Mol Imaging Biol 10, 349–355 (2008). https://doi.org/10.1007/s11307-008-0162-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11307-008-0162-3