Abstract
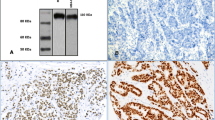
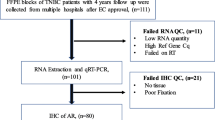
One of the active intracellular pathways/networks in triple-negative breast carcinoma (TNBC) is that of the androgen receptor (AR). In this study, we examined AR and androgen-metabolising enzyme immunoreactivity in subcategories of TNBC to further elucidate the roles of androgenic pathways in TNBC. We utilised formalin-fixed paraffin-embedded breast cancer samples from ductal carcinoma in situ (DCIS) and invasive ductal carcinoma patient cohorts. We then used immunohistochemistry to classify these samples into basal-like and non-basal samples and to assess interactions between AR, androgen-metabolising enzymes and proliferation. To further substantiate our hypothesis and provide mechanistic insights, we also looked at the expression and regulation of these factors in publically available microarray data and in a panel of TNBC AR-positive cell lines. DCIS was associated with higher levels of AR and enzymes (p < 0.02), although a similar difference was not noticed in basal and non-basal samples. AR and enzymes were correlated in all states. In TNBC cell lines (MDA-MD-453, MFM-223 and SUM185-PE), we found that DHT treatment up-regulated 5αR1 and 17βHSD5 suggesting a mechanistic explanation for the correlations observed in the histological samples. Publicly available microarray data in TNBC cases suggested similar patterns to those observed in histological samples. In the majority of settings, including publically available microarray data, an inverse association between AR and proliferation was detected. These findings suggest that decreases in AR and androgen-metabolising enzymes may be involved in the increased biological aggressiveness in TNBC development.






Similar content being viewed by others
Abbreviations
- 5αR1:
-
Five alpha reductase one
- 17βHSD5:
-
Seventeen beta hydroxysteroid dehydrogenase five
- AR:
-
Androgen receptor
- BL2:
-
Basal-like group two
- CK5/6:
-
Cytokeratin five/six
- DCIS:
-
Ductal carcinoma in situ
- EGFR:
-
Epidermal growth factor receptor
- EMT:
-
Epithelial to mesenchymal transition
- ER:
-
Oestrogen receptor
- FOXA1:
-
Forkhead box protein A1
- IDC:
-
Invasive ductal carcinoma
- LAR:
-
Luminal androgen receptor
- LI:
-
Labelling index
- mRNA:
-
Messenger ribonucleic acid
- MSL:
-
Mesenchymal stem like
- PCNA:
-
Proliferating cell nuclear antigen
- RPL13A:
-
Ribosomal protein L 13a
- TNBC:
-
Triple-negative breast cancer
References
Bosch A, Eroles P, Zaragoza R, Vina JR, Lluch A (2010) Triple-negative breast cancer: molecular features, pathogenesis, treatment and current lines of research. Cancer Treat Rev 36(3):206–215. doi:10.1016/j.ctrv.2009.12.002
Hickey TE, Robinson JL, Carroll JS, Tilley WD (2012) Minireview: the androgen receptor in breast tissues: growth inhibitor, tumor suppressor, oncogene? Mol Endocrinol 26(8):1252–1267. doi:10.1210/me.2012-1107
McNamara KM, Yoda T, Takagi K, Miki Y, Suzuki T, Sasano H (2013) Androgen receptor in triple negative breast cancer. J Steroid Biochem Mol Biol 133:66–76. doi:10.1016/j.jsbmb.2012.08.007
Farmer P, Bonnefoi H, Becette V, Tubiana-Hulin M, Fumoleau P, Larsimont D, Macgrogan G, Bergh J, Cameron D, Goldstein D, Duss S, Nicoulaz AL, Brisken C, Fiche M, Delorenzi M, Iggo R (2005) Identification of molecular apocrine breast tumours by microarray analysis. Oncogene 24(29):4660–4671. doi:10.1038/sj.onc.1208561
Ormandy CJ, Clarke CL, Kelly PA, Sutherland RL (1992) Androgen regulation of prolactin-receptor gene expression in MCF-7 and MDA-MB-453 human breast cancer cells. Int J Cancer 50(5):777–782
Hall RE, Tilley WD, McPhaul MJ, Sutherland RL (1992) Regulation of androgen receptor gene expression by steroids and retinoic acid in human breast-cancer cells. Int J Cancer 52(5):778–784
Birrell SN, Bentel JM, Hickey TE, Ricciardelli C, Weger MA, Horsfall DJ, Tilley WD (1995) Androgens induce divergent proliferative responses in human breast cancer cell lines. J Steroid Biochem Mol Biol 52(5):459–467
Bentel JM, Birrell SN, Pickering MA, Holds DJ, Horsfall DJ, Tilley WD (1999) Androgen receptor agonist activity of the synthetic progestin, medroxyprogesterone acetate, in human breast cancer cells. Mol Cell Endocrinol 154(1–2):11–20
Mitchell S, Abel P, Madaan S, Jeffs J, Chaudhary K, Stamp G, Lalani el N (2002) Androgen-dependent regulation of human MUC1 mucin expression. Neoplasia 4(1):9–18
Doane AS, Danso M, Lal P, Donaton M, Zhang L, Hudis C, Gerald WL (2006) An estrogen receptor-negative breast cancer subset characterized by a hormonally regulated transcriptional program and response to androgen. Oncogene 25(28):3994–4008. doi:10.1038/sj.onc.1209415
Robinson JL, MacArthur S, Ross-Innes CS, Tilley WD, Neal DE, Mills IG, Carroll JS (2011) Androgen receptor driven transcription in molecular apocrine breast cancer is mediated by FoxA1. EMBO J 30(15):3019–3027. doi:10.1038/emboj.2011.216
Peters AA, Buchanan G, Ricciardelli C, Bianco-Miotto T, Centenera MM, Harris JM, Jindal S, Segara D, Jia L, Moore NL, Henshall SM, Birrell SN, Coetzee GA, Sutherland RL, Butler LM, Tilley WD (2009) Androgen receptor inhibits estrogen receptor-alpha activity and is prognostic in breast cancer. Cancer Res 69(15):6131–6140. doi:10.1158/0008-5472.CAN-09-0452
Lehmann BD, Bauer JA, Chen X, Sanders ME, Chakravarthy AB, Shyr Y, Pietenpol JA (2011) Identification of human triple-negative breast cancer subtypes and preclinical models for selection of targeted therapies. J Clin Invest 121(7):2750–2767. doi:10.1172/JCI45014
Ni M, Chen Y, Lim E, Wimberly H, Bailey ST, Imai Y, Rimm DL, Liu XS, Brown M (2011) Targeting androgen receptor in estrogen receptor-negative breast cancer. Cancer Cell 20(1):119–131. doi:10.1016/j.ccr.2011.05.026
McNamara K, Yoda T, Miki Y, Chanplakorn N, Wongwaisayawan S, Incharoen P, Kongdan Y, Wang L, Takagi K, Mayu T, Nakamura Y, Suzuki T, Nemoto N, Miyashita M, Tamaki K, Ishida T, Ohuchi N, Sasano H (2013) Androgenic pathway in triple negative invasive ductal tumours: its correlation with tumour cell proliferation. Cancer Sci 104(5):639–646. doi:10.1111/cas.12121
Agoff SN, Swanson PE, Linden H, Hawes SE, Lawton TJ (2003) Androgen receptor expression in estrogen receptor-negative breast cancer. Immunohistochemical, clinical, and prognostic associations. Am J Clin Pathol 120(5):725–731. doi:10.1309/42F0-0D0D-JD0J-5EDT
Luo X, Shi YX, Li ZM, Jiang WQ (2010) Expression and clinical significance of androgen receptor in triple negative breast cancer. Chin J Cancer 29(6):585–590
Rakha EA, El-Sayed ME, Green AR, Lee AH, Robertson JF, Ellis IO (2007) Prognostic markers in triple-negative breast cancer. Cancer 109(1):25–32. doi:10.1002/cncr.22381
Labrie F, Luu-The V, Labrie C, Belanger A, Simard J, Lin SX, Pelletier G (2003) Endocrine and intracrine sources of androgens in women: inhibition of breast cancer and other roles of androgens and their precursor dehydroepiandrosterone. Endocr Rev 24(2):152–182
Allred DC (2010) Ductal carcinoma in situ: terminology, classification, and natural history. J Natl Cancer Inst Monogr 2010(41):134–138. doi:10.1093/jncimonographs/lgq035
Voduc KD, Cheang MC, Tyldesley S, Gelmon K, Nielsen TO, Kennecke H (2010) Breast cancer subtypes and the risk of local and regional relapse. J Clin Oncol 28(10):1684–1691. doi:10.1200/JCO.2009.24.9284
Cheang MC, Voduc D, Bajdik C, Leung S, McKinney S, Chia SK, Perou CM, Nielsen TO (2008) Basal-like breast cancer defined by five biomarkers has superior prognostic value than triple-negative phenotype. Clin Cancer Res 14(5):1368–1376. doi:10.1158/1078-0432.ccr-07-1658
Kennecke H, Yerushalmi R, Woods R, Cheang MC, Voduc D, Speers CH, Nielsen TO, Gelmon K (2010) Metastatic behavior of breast cancer subtypes. J Clin Oncol 28(20):3271–3277. doi:10.1200/JCO.2009.25.9820
Li J, Ding Z, Wang Z, Lu JF, Maity SN, Navone NM, Logothetis CJ, Mills GB, Kim J (2011) Androgen regulation of 5alpha-reductase isoenzymes in prostate cancer: implications for prostate cancer prevention. PLoS One 6(12):e28840. doi:10.1371/journal.pone.0028840
Schulze JJ, Karypidis H, Ekstrom L (2012) Basal and regulatory promoter studies of the AKR1C3 gene in relation to prostate cancer. Front Pharmacol 3:151. doi:10.3389/fphar.2012.00151
Asjoe FT, Altintas S, Huizing MT, Colpaert C, Marck EV, Vermorken JB, Tjalma WA (2007) The value of the Van Nuys Prognostic Index in ductal carcinoma in situ of the breast: a retrospective analysis. Breast J 13(4):359–367. doi:10.1111/j.1524-4741.2007.00443.x
Tamaki K, Ishida T, Tamaki N, Kamada Y, Uehara K, Miyashita M, Amari M, Tadano-Sato A, Takahashi Y, Watanabe M, McNamara K, Ohuchi N, Sasano H (2012) Analysis of clinically relevant values of Ki-67 labeling index in Japanese breast cancer patients. Breast Cancer. doi:10.1007/s12282-012-0387-5
Miyashita M, Ishida T, Ishida K, Tamaki K, Amari M, Watanabe M, Ohuchi N, Sasano H (2011) Histopathological subclassification of triple negative breast cancer using prognostic scoring system: five variables as candidates. Virchows Arch 458(1):65–72. doi:10.1007/s00428-010-1009-2
Coletta A, Molter C, Duque R, Steenhoff D, Taminau J, de Schaetzen V, Meganck S, Lazar C, Venet D, Detours V, Nowe A, Bersini H, Weiss Solis DY (2012) InSilico DB genomic datasets hub: an efficient starting point for analyzing genome-wide studies in GenePattern, Integrative Genomics Viewer, and R/Bioconductor. Genome Biol 13(11):R104. doi:10.1186/gb-2012-13-11-r104
Reich M, Liefeld T, Gould J, Lerner J, Tamayo P, Mesirov JP (2006) GenePattern 2.0. Nat Genet 38(5):500–501. doi:10.1038/ng0506-500
Chen X, Li J, Gray WH, Lehmann BD, Bauer JA, Shyr Y, Pietenpol JA (2012) TNBCtype: a subtyping tool for triple-negative breast cancer. Cancer Inform 11:147–156. doi:10.4137/CIN.S9983
Thike AA, Iqbal J, Cheok PY, Tse GM, Tan PH (2013) Ductal carcinoma in situ associated with triple negative invasive breast cancer: evidence for a precursor-product relationship. J Clin Pathol. doi:10.1136/jclinpath-2012-201428
Ishikawa Y, Horiguchi J, Toya H, Nakajima H, Hayashi M, Tagaya N, Takeyoshi I, Oyama T (2011) Triple-negative breast cancer: histological subtypes and immunohistochemical and clinicopathological features. Cancer Sci 102(3):656–662. doi:10.1111/j.1349-7006.2011.01858.x
Aguiar FN, Mendes HN, Bacchi CE, Carvalho FM (2013) Comparison of nuclear grade and immunohistochemical features in situ and invasive components of ductal carcinoma of breast. Rev Bras Ginecol Obstet 35(3):97–102
de Azambuja E, Cardoso F, de Castro G Jr, Colozza M, Mano, Durbecq V, Sotiriou C, Larsimont D, Piccart-Gebhart MJ, Paesmans M (2007) Ki-67 as prognostic marker in early breast cancer: a meta-analysis of published studies involving 12,155 patients. Br J Cancer 96(10):1504–1513. doi:10.1038/sj.bjc.6603756
Shibuya R, Suzuki T, Miki Y, Yoshida K, Moriya T, Ono K, Akahira J, Ishida T, Hirakawa H, Evans DB, Sasano H (2008) Intratumoral concentration of sex steroids and expression of sex steroid-producing enzymes in ductal carcinoma in situ of human breast. Endocr Relat Cancer 15(1):113–124. doi:10.1677/ERC-07-0092
Ni M, Chen Y, Fei T, Li D, Lim E, Liu XS, Brown M (2013) Amplitude modulation of androgen signaling by c-MYC. Genes Dev 27(7):734–748. doi:10.1101/gad.209569.112
Hackenberg R, Luttchens S, Hofmann J, Kunzmann R, Holzel F, Schulz KD (1991) Androgen sensitivity of the new human breast cancer cell line MFM-223. Cancer Res 51(20):5722–5727
Yeap BB, Krueger RG, Leedman PJ (1999) Differential posttranscriptional regulation of androgen receptor gene expression by androgen in prostate and breast cancer cells. Endocrinology 140(7):3282–3291
Gucalp A, Tolaney S, Isakoff SJ, Ingle JN, Liu MC, Carey LA, Blackwell K, Rugo H, Nabell L, Forero A, Stearns V, Doane AS, Danso M, Moynahan ME, Momen LF, Gonzalez JM, Akhtar A, Giri DD, Patil S, Feigin KN, Hudis CA, Traina TA, Translational Breast Cancer Research Consortium (2013) Phase II trial of bicalutamide in patients with androgen receptor-positive, estrogen receptor-negative metastatic Breast Cancer. Clin Cancer Res 19(19):5505–5512. doi:10.1158/1078-0432.CCR-12-3327
Acknowledgments
This study was in part supported by a research grant from Astra-Zeneca Japan. We would also like to acknowledge the Department of Pathology, Ramathibodi Hospital, Thailand for their kindness in sharing their TNBC IDC samples with us and the support from the technical staff at Tohoku University hospital and Mr. S. Nakamura for his valuable contribution to immunostaining slides. In addition, we would like to acknowledge the contribution of Mr. R. Stodart in proofreading of the manuscript text. Keely M. McNamara was supported in this work by a JSPS-AAS postdoctoral fellowship. Kristy A. Brown is supported by a NHMRC (Australia) Career Development Award GNT1007714.
Conflict of interest
The authors declare that they have no competing or conflicting interests.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
McNamara, K.M., Yoda, T., Nurani, A.M. et al. Androgenic pathways in the progression of triple-negative breast carcinoma: a comparison between aggressive and non-aggressive subtypes. Breast Cancer Res Treat 145, 281–293 (2014). https://doi.org/10.1007/s10549-014-2942-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-014-2942-6