Abstract
Introduction
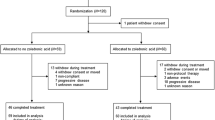
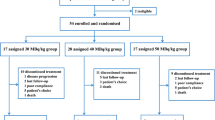
In this prospective study, the efficacy and safety of radiotherapy combined with zoledronic acid was evaluated.
Materials and methods
Breast cancer patients with painful bone metastases were randomized to either high- or reduced-dose radiotherapy. All patients received zoledronic acid (4 mg) every 28 days from the beginning of radiotherapy. Analgesic and pain scores in addition to visual analog score (VAS) for treatment satisfaction and whole-body bone scintigraphy were evaluated.
Results and conclusion
No significant differences could be found in analgesic or pain scores and bone scintigraphy results between the groups. Our results suggest that reduced-dose radiotherapy produces similar response rates and response durations when used concomitantly with zoledronic acid.





Similar content being viewed by others
References
Domchek SM, Younger J, Finkelstein DM, Seiden MW (2000) Predictors of skeletal complications in patients with metastatic breast carcinoma. Cancer 89:363–368. doi:10.1002/1097-0142(20000715)89:2<363::AID-CNCR22>3.0.CO;2-3
Lipton A (2005) Management of bone metastases in breast cancer. Curr Treat Options Oncol 6:161–171. doi:10.1007/s11864-005-0023-0
Coleman RE (2001) Metastatic bone disease: clinical features, pathophsiology and treatment strategies. Cancer Treat Rev 27:165–176. doi:10.1053/ctrv.2000.0210
Coleman RE, Rubens RD (1987) The clinical course of bone metastases from breast cancer. Br J Cancer 55:61–66
Bates T (1992) A review of local radiotherapy in the treatment of bone metastases and cord compression. Int J Radiat Oncol Biol Phys 23:217–221
Maher EJ (1992) The use of palliative radiotherapy in the management of breast cancer. Eur J Cancer 28:706–710. doi:10.1016/S0959-8049(05)80131-5
Fleisch H (2002) Development of bisphophonates. Breast Cancer Res 4:30–34. doi:10.1186/bcr414
Gnant M, Mlineritsch B, Schippinger W et al (2008) Adjuvant endocrine therapy plus zoledronic acid in premenopausal women with early-stage breast cancer: 5-year follow-up of the ABCSG-12 bone-mineral density substudy. Lancet Oncol 9:840–849. doi:10.1016/S1470-2045(08)70204-3
Hillner BE, Ingle JN, Chlebowski RT, Gralow J, Yee GC, Janjan NA, Cauley JA, Blumenstein BA, Albain KS, Lipton A, Brown S, American Society of Clinical Oncology (2003) 2003 update on the role of bisphosphonates and bone health issues in women with breast cancer. J Clin Oncol 21:4042–4057. doi:10.1200/JCO.2003.08.017
Price P, Hoskin PJ, Easton D, Austin D, Palmer SG, Easton D, Austin D, Palmer SG, Yarnold JR (1986) Prospective randomized trial of single and multifraction radiotherapy schedules in the treatment of painful bony metastases. Radiother Oncol 6:247–255. doi:10.1016/S0167-8140(86)80191-8
Tong D, Gillick L, Hendrickson FR (1982) The palliation of symptomatic osseous metastases. Final results of the study by the Radiation Therapy Oncology Group. Cancer 50:893–899. doi:10.1002/1097-0142(19820901)50:5<893::AID-CNCR2820500515>3.0.CO;2-Y
Madsen EL (1983) Painful bone metastases: efficacy of radiotherapy assessed by the patients: a randomized trial comparing 4 Gy × 6 versus 10 Gy × 2. Int J Radiat Oncol Biol Phys 9:1775–1779
Hartsell WF, Scott CB, Bruner DW, Scarantino CW, Ivker RA, Roach M, Suh JH, Demas WF, Movsas B, Petersen IA, Konski AA, Cleeland CS, Janjan NA, DeSilvio M (2005) Randomized trial of short- versus long- course radiotherapy for palliation of painful bone metastases. J Natl Cancer Inst 11:798–804
Wu JS, Wong R, Johnston M, Bezjak A, Whelan T (2003) Meta-analysis of dose-fractionation radiotherapy trials for the palliation of painful bone metastases. Int J Radiat Oncol Biol Phys 55:594–605. doi:10.1016/S0360-3016(02)04147-0
van den Hout WB, van der Linden YM, Steenland E, Wiggenraad RG, Kievit J, de Haes H, Leer JW (2003) Single- versus multipl- fraction radiotherapy in patients with painful bone metastases: cost-Utility analysis based on a randomized trial. J Natl Cancer Inst 95:222–229
Sze WM, Shelley MD, Held I, Wilt TJ, Mason MD (2003) Palliation of metastatic bone pain: single fraction versus multifraction radiotherapy- A systematic rewiev of randomized trials. Clin Oncol 15:345–352. doi:10.1016/S0936-6555(03)00113-4
Crellin AM, Marks A, Maher EJ (1989) Why don’t British radiotherapists give single fractions of radiotherapy for bone metastases. Clin Oncol 1:63–66. doi:10.1016/S0936-6555(89)80036-6
Flanagan AM, Chambers TJ (1991) Inhibition of bone resorption by bisphosphonates: interactions between bisphosphonates, osteoclasts, and bone. Calcif Tissue Int 49:407–415. doi:10.1007/BF02555852
Rogers MJ, Gordon S, Benford HL, Coxon FP, Luckman SP, Monkkonen J, Frith JC (2000) Cellular and molecular mechanisms of action of bisphosphonates. Cancer 88(Suppl):2961–2978. doi:10.1002/1097-0142(20000615)88:12+<2961::AID-CNCR12>3.0.CO;2-L
Dunford JE, Thompson K, Coxon FP, Luckman SP, Hahn FM, Poulter CD, Ebetino FH, Rogers MJ (2001) Structure-activity relationships for inhibition of farnesyl diphosphate synthase in vitro and inhibition of bone resorptio in vivo by nitrogen-containing bisphosphonates. J Pharmacol Exp Ther 296:235–242
Alakangas A, Selander K, Mulari M, Halleen J, Lehenkari P, Mönkkönen J, Salo J, Väänänen K (2002) Alendronate disturbs vesicular trafficking in osteoclasts. Calcif Tissue Int 70:40–47. doi:10.1007/s002230010047
Fisher JE, Rodan GA, Reszka AA (2000) In vivo effects of bisphosphonates on the osteoclast mevalonate pathway. Endocrinology 141:4793–4796. doi:10.1210/en.141.12.4793
Boyle WJ, Simonet WS, Lacey DL (2003) Osteoclast differentiation and activation. Nature 423:337–342. doi:10.1038/nature01658
Pavlakis N, Schmidt RL, Stockler M (2005) Bisphosphonates for breast cancer. Cochrane Database Syst Rev 3:CD003474
Rosen LS, Gordon D, Kaminski M, Howell A, Belch A, Mackey J, Apffelstaedt J, Hussein MA, Coleman RE, Reitsma DJ, Chen BL, Seaman JJ (2003) Long-term efficacy and safety of zoledronic acid compared with pamidronate disodium in the treatment of skeletal complications in patients with advanced multiple myeloma or breast carcinoma: a randomized, double-blind, multicenter, comparative trial. Cancer 98:1735–1744. doi:10.1002/cncr.11701
Rosen LS, Gordon DH, Dugan W Jr, Major P, Eisenberg PD, Provencher L, Kaminski M, Simeone J, Seaman J, Chen BL, Coleman RE (2004) Zoledronic acid is superior to pamidronate for the treatment of bone metastases in breast carcinoma patients with at least one osteolytic lesion. Cancer 100:36–43. doi:10.1002/cncr.11892
Kohno N, Aogi K, Minami H, Nakamura S, Asaga T, Iino Y, Watanabe T, Goessl C, Ohashi Y, Takashima S (2005) Zoledronic acid significantly reduces skeletal complications compared with placebo in Japanese women with bone metastases from breast cancer: a Randomized, Placebo-Controlled Trial. J Clin Oncol 23:3314–3321. doi:10.1200/JCO.2005.05.116
Coleman RE, Rosen LS, Gordon D, Major P, Kaminski M, Apffelstaedt J, Howell A, Chen B-L, Seaman JJ (2002) Zoledronic acid (4 mg) significantly reduces the relative risk of developing a skeletal–related event compared with pamidronate (90 mg) in patients with breast cancer and bone metastasis. Breast Cancer Res Treat 76(suppl.):355
Senaratne SG, Pirianov G, Mansi JL, Arnett TR, Colston KW (2000) Bisphophonates induce apoptosis in human breast cancer cell lines. Br J Cancer 82:1459–1468
Morony S, Capparelli C, Sarosi I, Lacey DL, Dunstan CR, Kostenuik PJ (2001) Osteoprotegrin inhibits osteolysis and decreases skeletal tumor burden in syngeneic and nude mouse models of experimental bone metastases. Cancer Res 61:4432–4436
van der Pluijm G, Vloedgraven H, van Beek E, van der Wee-Pals L, Löwik C, Papapoulos S (1996) Biphophonates inhibit the adhesion of breast cancer cells to bone matrices in vitro. J Clin Invest 98:698–705. doi:10.1172/JCI118841
Boissier S, Magnetto S, Frappart L, Cuzin B, Ebetino FH, Delmas PD, Clezardin P (1997) Biphophonates inhibit prostate and breast carcinoma cell adhesion to unmineralized and mineralized bone extracellular matrices. Cancer Res 57:3890–3894
Vogt U, Bielawski KP, Bosse U, Schlotter CM (2004) Breast tumour growth inhibition in vitro through the combination of cyclophosphamide/metotrexate/5-fluorouracil, epirubicin/cyclophosphamide, epirubicin/paclitaxel, and epirubicin/docetaxel with the bisphosphonates ibandronate and zoledronic acid. Oncol Rep 12:1109–1114
Jagdev SP, Coleman RE, Shipman CM, Rostami-H A, Croucher PI (2001) The bisphosphonate, zoledronic acid, induces apoptosis of breast cancer cells: evidence for synergy with paclitaxel. Br J Cancer 84:1126–1134. doi:10.1054/bjoc.2001.1727
Neville-Webbe HL, Rostami-Hodjegan A, Evans CA, Coleman RE, Holen I (2005) Sequence- and schedule-dependent enhancement of zoledronic acid induced apoptosis by doxorubicin in breast and prostate cancer cells. Int J Cancer 113:364–371. doi:10.1002/ijc.20602
Magnetto S, Boissier S, Delmas PD, Clezardin P (1999) Additive antitumor activities of taxoids in combination with the biphosphonate ibandronate against invasion and adhesion of human breast carcinoma cells to bone. Int J Cancer 83:263–269. doi:10.1002/(SICI)1097-0215(19991008)83:2<263::AID-IJC19>3.0.CO;2-T
Ural AU, Avcu F, Candir M, Guden M, Ozcan MA (2006) In vitro synergistic effects of zoledronic acid and radiation on breast cancer cells. Breast Cancer Res 8:R52. doi:10.1186/bcr1543
Arrington SA, Damron TA, Mann KA, Allen MJ (2008) Concurrent administration of zoledronic acid and irradiation leads to improved bone density, biomechanical strength, and microarchitecture in a mouse model of tumor-induced osteolysis. J Surg Oncol 97:284–290. doi:10.1002/jso.20949
Blitzer PH (1985) Reanalysis of the RTOG study of the palliation of symptomatic osseous metastasis. Cancer 55:1468–1472. doi:10.1002/1097-0142(19850401)55:7<1468::AID-CNCR2820550708>3.0.CO;2-M
Declaration of interest
Any actual or potential conflicts of interest do not exist.
Author information
Authors and Affiliations
Corresponding author
Additional information
Lale Atahan, Deceased
Rights and permissions
About this article
Cite this article
Atahan, L., Yıldız, F., Cengiz, M. et al. Zoledronic acid concurrent with either high- or reduced-dose palliative radiotherapy in the management of the breast cancer patients with bone metastases: a phase IV randomized clinical study. Support Care Cancer 18, 691–698 (2010). https://doi.org/10.1007/s00520-009-0663-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-009-0663-x