Abstract
Background
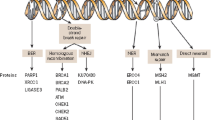
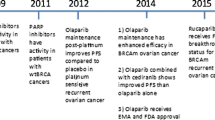
Ovarian cancer (OC) is the leading cause of death in women with gynecological cancer. Most patients are diagnosed at advanced stage with poor prognosis. Currently, surgical tumor debulking followed by chemotherapy based on platinum and taxane is the standard treatment for advanced OC. However, these patients remain at great risk for recurrence and developing drug resistance. Therefore, new treatment strategies are needed to improve outcomes for patients with advanced and recurrent OC. Olaparib (AZD2281, KU-0059436), as one of the best understood Poly-(ADP-ribose) polymerase (PARP) inhibitor targeting DNA repair mechanisms, caused more and more attention. Clinical trial data of Olaparib had been cumulated, which applied as the single-agent in relapsed OC monotherapy, especially for BRCA mutation associated OC.
Methods
In this review, we demonstrated the mechanism of PARP inhibitors and summarized clinical trial data and clinical development of Olaparib targeted OC in order to address a new promising therapy strategy for advanced relapsed OC.
Conclusion
Given the unprecedented clinical potential of Olaparib, the further research on Olaparib will have great significance in selection of OC patient populations that will respond to treatment.


Similar content being viewed by others
References
Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D (2011) Global cancer statistics. CA Cancer J Clin 61(2):69–90
Chi DS, Eisenhauer EL, Zivanovic O, Sonoda Y, Abu-Rustum NR, Levine DA, Guile MW, Bristow RE, Aghajanian C, Barakat RR (2009) Improved progression-free and overall survival in advanced ovarian cancer as a result of a change in surgical paradigm. Gynecol Oncol 114(1):26–31
Chambon P, Weill JD, Mandel P (1963) Nicotinamide mononucleotide activation of new DNA-dependent polyadenylic acid synthesizing nuclear enzyme. Biochem Biophys Res Commun 11:39–43
Langelier MF, Servent KM, Rogers EE, Pascal JM (2008) A third zinc-binding domain of human poly(ADP-ribose) polymerase-1 coordinates DNA-dependent enzyme activation. J Biol Chem 283(7):4105–4114
Tao Z, Gao P, Hoffman DW, Liu HW (2008) Domain C of human poly(ADP-ribose) polymerase-1 is important for enzyme activity and contains a novel zinc-ribbon motif. Biochemistry 47(21):5804–5813
Hassa PO, Haenni SS, Elser M, Hottiger MO (2006) Nuclear ADP-ribosylation reactions in mammalian cells: where are we today and where are we going? Microbiol Mol Biol Rev 70(3):789–829
de Murcia G, Schreiber V, Molinete M, Saulier B, Poch O, Masson M, Niedergang C (1994) Ménissier de Murcia J. Structure and function of poly(ADP-ribose) polymerase. Mol Cell Biochem 138(1–2):15–24
Rouleau M, Patel A, Hendzel MJ, Kaufmann SH, Poirier GG (2010) PARP inhibition: PARP1 and beyond. Nat Rev Cancer 10(4):293–301
Clark JB, Ferris GM, Pinder S (1971) Inhibition of nuclear NAD nucleosidase and poly ADP-ribose polymerase activity from rat liver by nicotinamide and 5′-methyl nicotinamide. Biochim Biophys Acta 238(1):82–85
Durkacz BW, Omidiji O, Gray DA, Shall S (1980) (ADP-ribose)n participates in DNA excision repair. Nature 283(5747):593–596
Weil MK, Chen AP (2011) PARP inhibitor treatment in ovarian and breast cancer. Curr Probl Cancer 35(1):7–50
Menear KA, Adcock C, Boulter R, Cockcroft XL, Copsey L, Cranston A et al (2008) 4-[3-(4-cyclopropanecarbonylpiperazine-1-carbonyl)-4-fluorobenzyl]-2H-phthalazin-1-one: a novel bioavailable inhibitor of poly(ADP-ribose) polymerase-1. J Med Chem 51(20):6581–6591
Fong PC, Boss DS, Yap TA, Tutt A, Wu P, Mergui-Roelvink M et al (2009) Inhibition of poly(ADP-ribose) polymerase in tumors from BRCA mutation carriers. N Engl J Med 361(2):123–134
Ledermann J, Harter P, Gourley C, Friedlander M, Vergote I, Rustin G et al (2012) Olaparib maintenance therapy in platinum-sensitive relapsed ovarian cancer. N Engl J Med 366(15):1382–1392
Kaye SB, Lubinski J, Matulonis U et al (2012) Phase II, open-label, randomized, multicenter study comparing the efficacy and safety of olaparib, a poly (ADP-ribose) polymerase inhibitor, and pegylated liposomal doxorubicin in patients with BRCA1 or BRCA2 mutations and recurrent ovarian cancer. J Clin Oncol 30(4):372–379
Gelmon KA, Tischkowitz M, Mackay H, Swenerton K, Robidoux A, Tonkin K et al (2011) Olaparib in patients with recurrent high-grade serous or poorly differentiated ovarian carcinoma or triple-negative breast cancer: a phase 2, multicentre, open-label, non-randomised study. Lancet Oncol 12(9):852–861
Yamamoto N, Nokihara H, Yamada Y et al (2012) A phase I, dose-finding and pharmacokinetic study of Olaparib (AZD2281) in Japanese patients with advanced solid tumours. Cancer Sci 103(3):504–509
Audeh MW, Carmichael J, Penson RT et al (2010) Oral poly(ADP-ribose) polymerase inhibitor olaparib in patients with BRCA1 or BRCA2 mutations and recurrent ovarian cancer: a proof-of-concept trial. Lancet 376(9737):245–251
Ashworth A (2008) A synthetic lethal therapeutic approach: poly(ADP) ribose polymerase inhibitors for the treatment of cancers deficient in DNA double-strand break repair. J Clin Oncol 26(22):3785–3790
Underhill C, Toulmonde M, Bonnefoi H (2011) A review of PARP inhibitors: from bench to bedside. Ann Oncol 22(2):268–279
Fong PC, Yap TA, Boss DS et al (2010) Poly (ADP)-ribose polymerase inhibition: frequent durable responses in BRCA carrier ovarian cancer correlating with platinum-free interval. J Clin Oncol 28:2512–2519
Gordon AN, Fleagle JT, Guthrie D et al (2001) Recurrent epithelial ovarian carcinoma: a randomized phase III study of pegylated liposomal doxorubicin versus topotecan. J Clin Oncol 19(14):3312–3322
Adams SF, Marsh EB, Elmasri W et al (2011) A high response rate to liposomal doxorubicin is seen among women with BRCA mutations treated for recurrent epithelial ovarian cancer. Gynecol Oncol 123(3):486–491
Marchetti C, Imperiale L, Gasparri ML, Palaia I, Pignata S, Boni T, Bellati F (2012) Benedetti Panici P. Olaparib, PARP1 inhibitor in ovarian cancer. Expert Opin Investig Drugs 21(10):1575–1584
Palma JP, Rodriguez LE, Bontcheva-Diaz VD et al (2008) The PARP inhibitor, ABT-888 potentiates temozolomide: correlation with drug levels and reduction in PARP activity in vivo. Anticancer Res 28(5A):2625–2635
Donawho CK, Luo Y, Luo Y et al (2007) ABT-888, an orally active poly (ADP-ribose) polymerase inhibitor that potentiates DNA-damaging agents in preclinical tumour models. Clin Cancer Res 13(9):2728–2737
Carden CP, Yap TA, Kaye SB (2010) PARP inhibition: targeting the Achilles’ heel of DNA repair to treat germline and sporadic ovarian cancers. Curr Opin Oncol 22(5):473–480
Samol J, Ranson M, Scott E et al (2011) Safety and tolerability of the poly(ADP-ribose) polymerase (PARP) inhibitor, Olaparib (AZD2281) in combination with topotecan for the treatment of patients with advanced solid tumours: a phase I study. Invest New Drugs 30(4):1493–1500
Khan OA, Gore M, Lorigan P et al (2011) A phase I study of the safety and tolerability of olaparib (AZD2281, KU0059436) and dacarbazine in patients with advanced solid tumours. Br J Cancer 104:750–755
Dent RA, Lindeman GJ, Clemons M et al (2011) Safety and efficacy of the oral PARP inhibitor Olaparib (AZD2281) in combination with paclitaxel for the first or second-line treatment of patients with metastatic triple-negative breast cancer: results from the safety cohort of a phase I/II multicenter trial. J Clin Oncol 28(15S):1018 (abstract)
Rajan A, Carter CA, Kelly RJ et al (2012) A phase I combination study of olaparib with cisplatin and gemcitabine in adults with solid tumours. Clin Cancer Res 18(8):2344–2351
Dean E, Middleton MR, Pwint T et al (2012) Phase I study to assess the safety and tolerability of Olaparib in combination with Bevacizumab in patients with advanced solid tumours. Br J Cancer 106(3):468–474
Chan N, Bristow RG (2010) Contextual synthetic lethality and/or loss of heterozygosity: tumour hypoxia and modification of DNA repair. Clin Cancer Res 16:4553–4560
Acknowledgments
This work was supported by Tianjin Health Bureau of Science and Technology Funds (2012KZ073).
Conflict of interest
None of the authors has any potential conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Y. Chen and L. Zhang contributed equally to this work.
Rights and permissions
About this article
Cite this article
Chen, Y., Zhang, L. & Hao, Q. Olaparib: a promising PARP inhibitor in ovarian cancer therapy. Arch Gynecol Obstet 288, 367–374 (2013). https://doi.org/10.1007/s00404-013-2856-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-013-2856-2