Abstract
Objectives
To evaluate the utility of perfusion MRI as a potential biomarker for predicting response to chemoradiotherapy (CRT) in locally advanced rectal cancer.
Methods
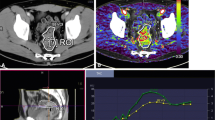
Thirty-nine patients with primary rectal carcinoma who were scheduled for preoperative CRT were prospectively recruited. Perfusion MRI was performed with a 3.0-T MRI system in all patients before therapy, at the end of the 2nd week of therapy, and before surgery. The K trans (volume transfer constant) and V e (extracellular extravascular space fraction) were calculated.
Results
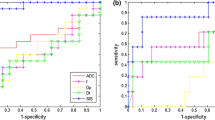
Before CRT, the mean tumour K trans in the downstaged group was significantly higher than that in the non-downstaged group (P = 0.0178), but there was no significant difference between tumour regression grade (TRG) responders and TRG non-responders (P = 0.1392). Repeated-measures analysis of variance (ANOVA) showed significant differences for evolution of K trans values both between downstaged and non-downstaged groups (P = 0.0215) and between TRG responders and TRG non-responders (P = 0.0001). Regarding V e, no significant differences were observed both between downstaged and non-downstaged groups (P = 0.689) or between TRG responders and TRG non-responders (P = 0.887).
Conclusion
Perfusion MRI of rectal cancer can be useful for assessing tumoural K trans changes by CRT. Tumours with high pre-CRT K trans values tended to respond favourably to CRT, particularly in terms of downstaging criteria.
Key Points
• Perfusion MRI can now assess therapeutic response of tumours to therapy.
• Tumours with high initial K trans values responded favourably to chemoradiotherapy.
• Perfusion MRI of rectal cancer may help with decisions about management.



Similar content being viewed by others
References
Padhani AR, Liu G, Koh DM et al (2009) Diffusion-weighted magnetic resonance imaging as a cancer biomarker: consensus and recommendations. Neoplasia 11:102–125
Gray LH, Conger AD, Ebert M, Hornsey S, Scott OC (1953) The concentration of oxygen dissolved in tissues at the time of irradiation as a factor in radiotherapy. Br J Radiol 26:638–648
Overgaard J, Horsman MR (1996) Modification of hypoxia-induced radioresistance in tumors by the use of oxygen and sensitizers. Semin Radiat Oncol 6:10–21. doi:10.1053/SRAO0060010
Wolmark N, Piedbois P, Rougier P et al (1998) Efficacy of intravenous continuous infusion of fluorouracil compared with bolus administration in advanced colorectal cancer. J Clin Oncol 16:301–308
Vaupel P, Kallinowski F, Okunieff P (1989) Blood flow, oxygen and nutrient supply, and metabolic microenvironment of human tumors: a review. Cancer Res 49:6449–6465
Jain RK (2005) Normalization of tumor vasculature: an emerging concept in antiangiogenic therapy. Science 307:58–62. doi:10.1126/science.1104819
Tofts PS, Brix G, Buckley DL et al (1999) Estimating kinetic parameters from dynamic contrast-enhanced T(1)-weighted MRI of a diffusable tracer: standardized quantities and symbols. J Magn Reson Imaging 10:223–232
Janssen MH, Aerts HJ, Kierkels RG et al (2010) Tumor perfusion increases during hypofractionated short-course radiotherapy in rectal cancer: sequential perfusion-CT findings. Radiother Oncol 94:156–160. doi:10.1016/j.radonc.2009.12.013
Bellomi M, Petralia G, Sonzogni A, Zampino MG, Rocca A (2007) CT perfusion for the monitoring of neoadjuvant chemotherapy and radiation therapy in rectal carcinoma: initial experience. Radiology 244:486–493. doi:10.1148/radiol.2442061189
Sahani DV, Kalva SP, Hamberg LM et al (2005) Assessing tumor perfusion and treatment response in rectal cancer with multisection CT: initial observations. Radiology 234:785–792. doi:10.1148/radiol.2343040286
Tofts PS (1997) Modeling tracer kinetics in dynamic Gd-DTPA MR imaging. J Magn Reson Imaging 7:91–101
George ML, Dzik-Jurasz AS, Padhani AR et al (2001) Non-invasive methods of assessing angiogenesis and their value in predicting response to treatment in colorectal cancer. Br J Surg 88:1628–1636. doi:10.1046/j.0007-1323.2001.01947.x
Tofts PS, Kermode AG (1991) Measurement of the blood-brain barrier permeability and leakage space using dynamic MR imaging. 1. Fundamental concepts. Magn Reson Med 17:357–367
Weinmann HJ, Laniado M, Mutzel W (1984) Pharmacokinetics of GdDTPA/dimeglumine after intravenous injection into healthy volunteers. Physiol Chem Phys Med NMR 16:167–172
Mandard AM, Dalibard F, Mandard JC et al (1994) Pathologic assessment of tumor regression after preoperative chemoradiotherapy of esophageal carcinoma. Clinicopathologic correlations. Cancer 73:2680–2686
Vecchio FM, Valentini V, Minsky BD et al (2005) The relationship of pathologic tumor regression grade (TRG) and outcomes after preoperative therapy in rectal cancer. Int J Radiat Oncol Biol Phys 62:752–760. doi:10.1016/j.ijrobp.2004.11.017
Cooper RA, Carrington BM, Loncaster JA et al (2000) Tumour oxygenation levels correlate with dynamic contrast-enhanced magnetic resonance imaging parameters in carcinoma of the cervix. Radiother Oncol 57:53–59
Janjan NA, Crane C, Feig BW et al (2001) Improved overall survival among responders to preoperative chemoradiation for locally advanced rectal cancer. Am J Clin Oncol 24:107–112
Sun YS, Zhang XP, Tang L et al (2010) Locally advanced rectal carcinoma treated with preoperative chemotherapy and radiation therapy: preliminary analysis of diffusion-weighted MR imaging for early detection of tumor histopathologic downstaging. Radiology 254:170–178. doi:10.1148/radiol.2541082230
Mayr NA, Yuh WT, Magnotta VA et al (1996) Tumor perfusion studies using fast magnetic resonance imaging technique in advanced cervical cancer: a new noninvasive predictive assay. Int J Radiat Oncol Biol Phys 36:623–633
Harvey C, Dooher A, Morgan J, Blomley M, Dawson P (1999) Imaging of tumour therapy responses by dynamic CT. Eur J Radiol 30:221–226
Wheeler JM, Warren BF, Jones AC, Mortensen NJ (1999) Preoperative radiotherapy for rectal cancer: implications for surgeons, pathologists and radiologists. Br J Surg 86:1108–1120. doi:10.1046/j.1365-2168.1999.01209.x
Dworak O, Keilholz L, Hoffmann A (1997) Pathological features of rectal cancer after preoperative radiochemotherapy. Int J Colorectal Dis 12:19–23
Acknowledgments
This study was supported by a grant from the Korea Healthcare Technology R&D Project, Ministry of Health Welfare, Republic of Korea (A070001).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lim, J.S., Kim, D., Baek, SE. et al. Perfusion MRI for the prediction of treatment response after preoperative chemoradiotherapy in locally advanced rectal cancer. Eur Radiol 22, 1693–1700 (2012). https://doi.org/10.1007/s00330-012-2416-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-012-2416-3