Abstract
Background
While surgical resection of pancreatic adenocarcinoma provides the only chance of cure, long-term survival remains poor. Immunotherapy may improve outcomes, especially as adjuvant to local therapies. Gene-mediated cytotoxic immunotherapy (GMCI) generates a systemic anti-tumor response through local delivery of an adenoviral vector expressing the HSV-tk gene (aglatimagene besadenovec, AdV-tk) followed by anti-herpetic prodrug. GMCI has demonstrated synergy with standard of care (SOC) in other tumor types. This is the first application in pancreatic cancer.
Methods
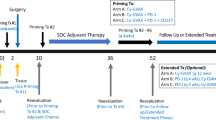
Four dose levels (3 × 1010 to 1 × 1012 vector particles) were evaluated as adjuvant to surgery for resectable disease (Arm A) or to 5-FU chemoradiation for locally advanced disease (Arm B). Each patient received two cycles of AdV-tk + prodrug.
Results
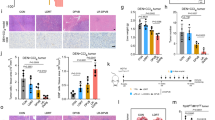
Twenty-four patients completed therapy, 12 per arm, with no dose-limiting toxicities. All Arm A patients were explored, eight were resected, one was locally advanced and three had distant metastases. CD8+ T cell infiltration increased an average of 22-fold (range sixfold to 75-fold) compared with baseline (p = 0.0021). PD-L1 expression increased in 5/7 samples analyzed. One node-positive resected patient is alive >66 months without recurrence. Arm B RECIST response rate was 25 % with a median OS of 12 months and 1-year survival of 50 %. Patient-reported quality of life showed no evidence of deterioration.
Conclusions
AdV-tk can be safely combined with pancreatic cancer SOC without added toxicity. Response and survival compare favorably to expected outcomes and immune activity increased. These results support further evaluation of GMCI with more modern chemoradiation and surgery as well as PD-1/PD-L1 inhibitors in pancreatic cancer.


Similar content being viewed by others
Abbreviations
- AdV-tk:
-
Adenoviral vector expressing the HSV-thymidine kinase gene
- AEs:
-
Adverse events
- APCs:
-
Antigen-presenting cells
- CTCAE:
-
Common Terminology Criteria for Adverse Events
- DLTs:
-
Dose-limiting toxicities
- EUS:
-
Endoscopic ultrasonography
- FACT-Hep:
-
Functional Assessment of Cancer Therapy-Hepatobiliary
- GMCI:
-
Gene-mediated cytotoxic immunotherapy
- LAPC:
-
Locally advanced pancreatic cancer
- MDSCs:
-
Myeloid-derived suppressor cells
- OS:
-
Overall survival
- PanCa:
-
Pancreatic cancer
- PD-1:
-
Programmed death 1
- PD-L1:
-
Programmed death ligand 1
- PFS:
-
Progression-free survival
- PR:
-
Partial response
- PD:
-
Progressive disease
- RECIST:
-
Response Evaluation Criteria in Solid Tumors
- RT:
-
Radiation therapy
- SD:
-
Stable disease
- SOC:
-
Standard of care
- TK:
-
Thymidine kinase protein from the HSV-thymidine kinase gene
- T-regs:
-
Regulatory T cells
References
Hidalgo M (2010) Pancreatic cancer. N Engl J Med 362:1605–1617. doi:10.1056/NEJMra0901557
Conroy T, Desseigne F, Ychou M, Bouché O, Guimbaud R, Bécouarn Y, Adenis A, Raoul J-L, Gourgou-Bourgade S, de la Fouchardière C, Bennouna J, Bachet J-B, Khemissa-Akouz F, Péré-Vergé D, Delbaldo C, Assenat E, Chauffert B, Michel P, Montoto-Grillot C, Ducreux M (2011) FOLFIRINOX versus gemcitabine for metastatic pancreatic cancer. N Engl J Med 364:1817–1825. doi:10.1056/NEJMoa1011923
Von-Hoff DD, Ervin T, Arena FP, Chiorean EG, Infante J, Moore M, Seay T, Tjulandin SA, Ma WW, Saleh MN, Harris M, Reni M, Dowden S, Laheru D, Bahary N, Ramanathan RK, Tabernero J, Hidalgo M, Goldstein D, Van Cutsem E, Wei X, Iglesias J, Renschler MF (2013) Increased survival in pancreatic cancer with nab-paclitaxel plus gemcitabine. N Engl J Med 369:1691–1703. doi:10.1056/NEJMoa1304369
Aguilar LK, Guzik BW, Aguilar-Cordova E (2011) Cytotoxic immunotherapy strategies for cancer: mechanisms and clinical development. J Cell Biochem 112:1969–1977
Rosenfeld ME, Vickers SM, Raben D, Wang M, Sampson L, Feng M, Jaffee E, Curiel DT (1997) Pancreatic carcinoma cell killing via adenoviral mediated delivery of the herpes simplex virus thymidine kinase gene. Ann Surg 225:609–618 discussion 618–620
Hall SJ, Mutchnik SE, Chen SH, Woo SL, Thompson TC (1997) Adenovirus-mediated herpes simplex virus thymidine kinase gene and ganciclovir therapy leads to systemic activity against spontaneous and induced metastasis in an orthotopic mouse model of prostate cancer. Int J Cancer 70:183–187
Perez-Cruet MJ, Trask TW, Chen SH, Goodman JC, Woo SL, Grossman RG, Shine HD (1994) Adenovirus-mediated gene therapy of experimental gliomas. J Neurosci Res 39:506–511. doi:10.1002/jnr.490390417
Vile RG, Nelson JA, Castleden S, Chong H, Hart IR (1994) Systemic gene therapy of murine melanoma using tissue specific expression of the HSVtk gene involves an immune component. Cancer Res 54:6228–6234
Agard C, Ligeza C, Dupas B, Izembart A, El Kouri C, Moullier P, Ferry N (2001) Immune-dependent distant bystander effect after adenovirus-mediated suicide gene transfer in a rat model of liver colorectal metastasis. Cancer Gene Ther 8:128–136. doi:10.1038/sj.cgt.7700281
Predina JD, Judy B, Aliperti LA, Fridlender ZG, Blouin A, Kapoor V, Laguna B, Nakagawa H, Rustgi AK, Aguilar L, Aguilar-Cordova E, Albelda SM, Singhal S (2011) Neoadjuvant in situ gene-mediated cytotoxic immunotherapy improves postoperative outcomes in novel syngeneic esophageal carcinoma models. Cancer Gene Ther 18:871–883. doi:10.1038/cgt.2011.56
Gagandeep S, Brew R, Green B, Christmas SE, Klatzmann D, Poston GJ, Kinsella AR (1996) Prodrug-activated gene therapy: involvement of an immunological component in the “bystander effect”. Cancer Gene Ther 3:83–88
Kuriyama S, Kikukawa M, Masui K, Okuda H, Nakatani T, Akahane T, Mitoro A, Tominaga K, Tsujinoue H, Yoshiji H, Okamoto S, Fukui H, Ikenaka K (1999) Cancer gene therapy with HSV-tk/GCV system depends on T-cell-mediated immune responses and causes apoptotic death of tumor cells in vivo. Int J Cancer 83:374–380
Sukin SW, Chhikara M, Zhu X, Ayala G, Aguilar LK, O’Brian Smith E, Miles BJ, Thompson TC, Kadmon D, Aguilar-Cordova E (2001) In vivo surgical resection plus adjuvant gene therapy in the treatment of mammary and prostate cancer. Mol Ther 3:500–506. doi:10.1006/mthe.2001.0285
Predina JD, Kapoor V, Judy BF, Cheng G, Fridlender ZG, Albelda SM, Singhal S (2012) Cytoreduction surgery reduces systemic myeloid suppressor cell populations and restores intratumoral immunotherapy effectiveness. J Hematol Oncol 5:34. doi:10.1186/1756-8722-5-34
Chhikara M, Huang H, Vlachaki MT, Zhu X, Teh B, Chiu KJ, Woo S, Berner B, Smith EO, Oberg KC, Aguilar LK, Thompson TC, Butler EB, Aguilar-Cordova E (2001) Enhanced therapeutic effect of HSV-tk+ GCV gene therapy and ionizing radiation for prostate cancer. Mol Ther 3:536–542. doi:10.1006/mthe.2001.0298
Formenti SC, Demaria S (2009) Systemic effects of local radiotherapy. Lancet Oncol 10:718–726. doi:10.1016/S1470-2045(09)70082-8
Boucher PD, Shewach DS (2005) In vitro and in vivo enhancement of ganciclovir-mediated bystander cytotoxicity with gemcitabine. Mol Ther 12:1064–1071. doi:10.1016/j.ymthe.2005.07.643
Wildner O, Blaese RM, Candotti F (1999) Enzyme prodrug gene therapy: synergistic use of the herpes simplex virus-cellular thymidine kinase/ganciclovir system and thymidylate synthase inhibitors for the treatment of colon cancer. Cancer Res 59:5233–5238
Fridlender ZG, Sun J, Singhal S, Kapoor V, Cheng G, Suzuki E, Albelda SM (2010) Chemotherapy delivered after viral immunogene therapy augments antitumor efficacy via multiple immune-mediated mechanisms. Mol Ther 18:1947–1959. doi:10.1038/mt.2010.159
Vincent J, Mignot G, Chalmin F, Ladoire S, Bruchard M, Chevriaux A, Martin F, Apetoh L, Rébé C, Ghiringhelli F (2010) 5-Fluorouracil selectively kills tumor-associated myeloid-derived suppressor cells resulting in enhanced T cell-dependent antitumor immunity. Cancer Res 70:3052–3061. doi:10.1158/0008-5472.CAN-09-3690
Shevchenko I, Karakhanova S, Soltek S, Link J, Bayry J, Werner J, Umansky V, Bazhin AV (2013) Low-dose gemcitabine depletes regulatory T cells and improves survival in the orthotopic Panc02 model of pancreatic cancer. Int J Cancer 133:98–107. doi:10.1002/ijc.27990
Yanagimoto H, Takai S, Satoi S, Toyokawa H, Takahashi K, Terakawa N, Kwon A-H, Kamiyama Y (2005) Impaired function of circulating dendritic cells in patients with pancreatic cancer. Clin Immunol 114:52–60. doi:10.1016/j.clim.2004.09.007
Liyanage UK, Moore TT, Joo H-G, Tanaka Y, Herrmann V, Doherty G, Drebin JA, Strasberg SM, Eberlein TJ, Goedegebuure PS, Linehan DC (2002) Prevalence of regulatory T cells is increased in peripheral blood and tumor microenvironment of patients with pancreas or breast adenocarcinoma. J Immunol 169:2756–2761
Ayala G, Satoh T, Li R, Shalev M, Gdor Y, Aguilar-Cordova E, Frolov A, Wheeler TM, Miles BJ, Rauen K, Teh BS, Butler EB, Thompson TC, Kadmon D (2006) Biological response determinants in HSV-tk+ ganciclovir gene therapy for prostate cancer. Mol Ther 13:716–728. doi:10.1016/j.ymthe.2005.11.022
Miles BJ, Shalev M, Aguilar-Cordova E, Timme TL, Lee HM, Yang G, Adler HL, Kernen K, Pramudji CK, Satoh T, Gdor Y, Ren C, Ayala G, Wheeler TM, Butler EB, Kadmon D, Thompson TC (2001) Prostate-specific antigen response and systemic T cell activation after in situ gene therapy in prostate cancer patients failing radiotherapy. Hum Gene Ther 12:1955–1967. doi:10.1089/104303401753204535
Trask TW, Trask RP, Aguilar-Cordova E, Shine HD, Wyde PR, Goodman JC, Hamilton WJ, Rojas-Martinez A, Chen SH, Woo SL, Grossman RG (2000) Phase I study of adenoviral delivery of the HSV-tk gene and ganciclovir administration in patients with current malignant brain tumors. Mol Ther 1:195–203
Chévez-Barrios P, Chintagumpala M, Mieler W, Paysse E, Boniuk M, Kozinetz C, Hurwitz MY, Hurwitz RL (2005) Response of retinoblastoma with vitreous tumor seeding to adenovirus-mediated delivery of thymidine kinase followed by ganciclovir. J Clin Oncol 23:7927–7935. doi:10.1200/JCO.2004.00.1883
Sterman DH, Recio A, Vachani A, Sun J, Cheung L, DeLong P, Amin KM, Litzky LA, Wilson JM, Kaiser LR, Albelda SM (2005) Long-term follow-up of patients with malignant pleural mesothelioma receiving high-dose adenovirus herpes simplex thymidine kinase/ganciclovir suicide gene therapy. Clin Cancer Res 11:7444–7453
Hasenburg A, Tong XW, Fischer DC, Rojas-Martinez A, Nyberg-Hoffman C, Kaplan AL, Kaufman RH, Ramzy I, Aguilar-Cordova E, Kieback DG (2001) Adenovirus-mediated thymidine kinase gene therapy in combination with topotecan for patients with recurrent ovarian cancer: 2.5-year follow-up. Gynecol Oncol 83:549–554
Chiocca EA, Aguilar LK, Bell SD, Kaur B, Hardcastle J, Cavaliere R, McGregor J, Lo S, Ray-Chaudhuri A, Chakravarti A, Grecula J, Newton H, Harris KS, Grossman RG, Trask TW, Baskin DS, Monterroso C, Manzanera AG, Aguilar-Cordova E, New PZ (2011) Phase IB study of gene-mediated cytotoxic immunotherapy adjuvant to up-front surgery and intensive timing radiation for malignant glioma. J Clin Oncol 29:3611–3619. doi:10.1200/JCO.2011.35.5222
Rojas-Martínez A, Manzanera AG, Sukin SW, Esteban-María J, González-Guerrero JF, Gomez-Guerra L, Garza-Guajardo R, Flores-Gutiérrez JP, Elizondo Riojas G, Delgado-Enciso I, Ortiz-López R, Aguilar LK, Butler EB, Barrera-Saldaña HA, Aguilar-Cordova E (2013) Intraprostatic distribution and long-term follow-up after AdV-tk immunotherapy as neoadjuvant to surgery in patients with prostate cancer. Cancer Gene Ther 20:642–649. doi:10.1038/cgt.2013.56
Teh BS, Ayala G, Aguilar L, Mai W-Y, Timme TL, Vlachaki MT, Miles B, Kadmon D, Wheeler T, Caillouet J, Davis M, Carpenter LS, Lu HH, Chiu JK, Woo SY, Thompson T, Aguilar-Cordova E, Butler EB (2004) Phase I-II trial evaluating combined intensity-modulated radiotherapy and in situ gene therapy with or without hormonal therapy in treatment of prostate cancer-interim report on PSA response and biopsy data. Int J Radiat Oncol Biol Phys 58:1520–1529. doi:10.1016/j.ijrobp.2003.09.083
Chen SH, Shine HD, Goodman JC, Grossman RG, Woo SL (1994) Gene therapy for brain tumors: regression of experimental gliomas by adenovirus-mediated gene transfer in vivo. Proc Natl Acad Sci USA 91:3054–3057
Shirley LA, Aguilar LK, Aguilar-Cordova E, Bloomston M, Walker JP (2013) Therapeutic endoscopic ultrasonography: intratumoral injection for pancreatic adenocarcinoma. Gastroenterol Res Pract 2013:207129. doi:10.1155/2013/207129
Heffernan N, Cella D, Webster K, Odom L, Martone M, Passik S, Bookbinder M, Fong Y, Jarnagin W, Blumgart L (2002) Measuring health-related quality of life in patients with hepatobiliary cancers: the functional assessment of cancer therapy-hepatobiliary questionnaire. J Clin Oncol 20:2229–2239
Moore MJ, Goldstein D, Hamm J, Figer A, Hecht JR, Gallinger S, Au HJ, Murawa P, Walde D, Wolff RA, Campos D, Lim R, Ding K, Clark G, Voskoglou-Nomikos T, Ptasynski M, Parulekar W (2007) Erlotinib plus gemcitabine compared with gemcitabine alone in patients with advanced pancreatic cancer: a phase III trial of the National Cancer Institute of Canada Clinical Trials Group. J Clin Oncol 25:1960–1966. doi:10.1200/JCO.2006.07.9525
Crane CH, Abbruzzese JL, Evans DB, Wolff RA, Ballo MT, Delclos M, Milas L, Mason K, Charnsangavej C, Pisters PWT, Lee JE, Lenzi R, Vauthey JN, Wong ABS, Phan T, Nguyen Q, Janjan NA (2002) Is the therapeutic index better with gemcitabine-based chemoradiation than with 5-fluorouracil-based chemoradiation in locally advanced pancreatic cancer? Int J Radiat Oncol Biol Phys 52:1293–1302
Loehrer PJ, Feng Y, Cardenes H, Wagner L, Brell JM, Cella D, Flynn P, Ramanathan RK, Crane CH, Alberts SR, Benson AB (2011) Gemcitabine alone versus gemcitabine plus radiotherapy in patients with locally advanced pancreatic cancer: an Eastern Cooperative Oncology Group trial. J Clin Oncol 29:4105–4112. doi:10.1200/JCO.2011.34.8904
Katz MHG, Fleming JB, Lee JE, Pisters PWT (2010) Current status of adjuvant therapy for pancreatic cancer. Oncologist 15:1205–1213. doi:10.1634/theoncologist.2010-0121
Bekaii-Saab T, Goldberg RM (2013) FOLFIRINOX in locally advanced pancreas adenocarcinoma: back to the future? Oncologist 18:487–489. doi:10.1634/theoncologist.2013-0157
Faris JE, Blaszkowsky LS, McDermott S, Guimaraes AR, Szymonifka J, Huynh MA, Ferrone CR, Wargo JA, Allen JN, Dias LE, Kwak EL, Lillemoe KD, Thayer SP, Murphy JE, Zhu AX, Sahani DV, Wo JY, Clark JW, Fernandez-del Castillo C, Ryan DP, Hong TS (2013) FOLFIRINOX in locally advanced pancreatic cancer: the Massachusetts General Hospital Cancer Center experience. Oncologist 18:543–548. doi:10.1634/theoncologist.2012-0435
Ebrahimi B, Tucker SL, Li D, Abbruzzese JL, Kurzrock R (2004) Cytokines in pancreatic carcinoma: correlation with phenotypic characteristics and prognosis. Cancer 101:2727–2736. doi:10.1002/cncr.20672
Gabitass RF, Annels NE, Stocken DD, Pandha HA, Middleton GW (2011) Elevated myeloid-derived suppressor cells in pancreatic, esophageal and gastric cancer are an independent prognostic factor and are associated with significant elevation of the Th2 cytokine interleukin-13. Cancer Immunol Immunother 60:1419–1430. doi:10.1007/s00262-011-1028-0
Tseng JF, Willett CG, Fernandez-del Castillo C, Ryan DP, Clark JW, Zhu AX, Rattner DW, Winkelmann JL, Warshaw AL (2005) Patients undergoing treatment for pancreatic adenocarcinoma can mount an effective immune response to vaccinations. Pancreatology 5:67–74. doi:10.1159/000084492
Thomas AM, Santarsiero LM, Lutz ER, Armstrong TD, Chen Y-C, Huang L-Q, Laheru DA, Goggins M, Hruban RH, Jaffee EM (2004) Mesothelin-specific CD8(+) T cell responses provide evidence of in vivo cross-priming by antigen-presenting cells in vaccinated pancreatic cancer patients. J Exp Med 200:297–306
Laheru D, Lutz E, Burke J, Biedrzycki B, Solt S, Onners B, Tartakovsky I, Nemunaitis J, Le D, Sugar E, Hege K, Jaffee E (2008) Allogeneic granulocyte macrophage colony-stimulating factor-secreting tumor immunotherapy alone or in sequence with cyclophosphamide for metastatic pancreatic cancer: a pilot study of safety, feasibility, and immune activation. Clin Cancer Res 14:1455–1463. doi:10.1158/1078-0432.CCR-07-0371
Kaufman HL, Kim-Schulze S, Manson K, DeRaffele G, Mitcham J, Seo KS, Kim DW, Marshall J (2007) Poxvirus-based vaccine therapy for patients with advanced pancreatic cancer. J Transl Med 5:60. doi:10.1186/1479-5876-5-60
Wedén S, Klemp M, Gladhaug IP, Møller M, Eriksen JA, Gaudernack G, Buanes T (2011) Long-term follow-up of patients with resected pancreatic cancer following vaccination against mutant K-ras. Int J Cancer 128:1120–1128. doi:10.1002/ijc.25449
Sampson JH, Heimberger AB, Archer GE, Aldape KD, Friedman AH, Friedman HS, Gilbert MR, Herndon JE, McLendon RE, Mitchell DA, Reardon DA, Sawaya R, Schmittling RJ, Shi W, Vredenburgh JJ, Bigner DD (2010) Immunologic escape after prolonged progression-free survival with epidermal growth factor receptor variant III peptide vaccination in patients with newly diagnosed glioblastoma. J Clin Oncol 28:4722–4729. doi:10.1200/JCO.2010.28.6963
Lutz E, Yeo CJ, Lillemoe KD, Biedrzycki B, Kobrin B, Herman J, Sugar E, Piantadosi S, Cameron JL, Solt S, Onners B, Tartakovsky I, Choi M, Sharma R, Illei PB, Hruban RH, Abrams RA, Le D, Jaffee E, Laheru D (2011) A lethally irradiated allogeneic granulocyte-macrophage colony stimulating factor-secreting tumor vaccine for pancreatic adenocarcinoma. A Phase II trial of safety, efficacy, and immune activation. Ann Surg 253:328–335. doi:10.1097/SLA.0b013e3181fd271c
Pardoll DM (2012) The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer 12:252–264. doi:10.1038/nrc3239
Lutz ER, Wu AA, Bigelow E, Sharma R, Mo G, Soares K, Solt S, Dorman A, Wamwea A, Yager A, Laheru D, Wolfgang CL, Wang J, Hruban RH, Anders RA, Jaffee EM, Zheng L (2014) Immunotherapy converts nonimmunogenic pancreatic tumors into immunogenic foci of immune regulation. Cancer Immunol Res 2:616–631. doi:10.1158/2326-6066.CIR-14-0027
Blazer M, Wu C, Goldberg RM, Phillips G, Schmidt C, Muscarella P, Wuthrick E, Williams TM, Reardon J, Christopher Ellison E, Bloomston M, Bekaii-Saab T (2014) Neoadjuvant modified (m) FOLFIRINOX for locally advanced unresectable (LAPC) and borderline resectable (BRPC) adenocarcinoma of the pancreas. Ann Surg Oncol. doi:10.1245/s10434-014-4225-1
Acknowledgments
This work was supported in part by the National Cancer Institute at the National Institute of Health [Grant Number R43CA119847] and from The Ohio State University Comprehensive Cancer Center Core Grant from the National Institute of Health [P30CA016058]. The trial is listed in the ClinicalTrials.gov registry, clinical trials identification number NCT00638612.
Conflict of interest
L. K. Aguilar, D. Sanchez, A. G. Manzanera and E. Aguilar-Cordova are employees of Advantagene, Inc. All remaining authors have declared no conflicts of interest related to this study.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Aguilar, L.K., Shirley, L.A., Chung, V.M. et al. Gene-mediated cytotoxic immunotherapy as adjuvant to surgery or chemoradiation for pancreatic adenocarcinoma. Cancer Immunol Immunother 64, 727–736 (2015). https://doi.org/10.1007/s00262-015-1679-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-015-1679-3