Abstract
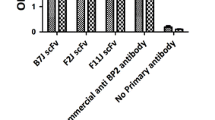
Prostate cancer is a major cause of death in older men, and bone metastasis is the primary cause of morbidity and mortality in prostate cancer. Prostate is an abundant source of nerve growth factor (NGF) that is secreted by malignant epithelial cells and utilized as an important autocrine factor for growth and metastasis. We previously showed that intravenous gammaglobulin (IVIg) contains natural antibodies against NGF, which inhibit growth and differentiation of the NGF-dependent cell line PC-12. In the present study, we examined the effects of these natural antibodies on in vitro migration or metastasis of two prostate cancer cell lines namely DU-145 and PC-3. Cancer cell migration was assessed using these cell lines in the upper chambers of Matrigel invasion chambers. The effects of IVIg and affinity-purified anti-NGF antibodies on cell migration through membrane into the lower chamber were assessed in dose/response experiments by a colorimetric method. Affinity-purified natural IgG anti-NGF antibody inhibited DU-145 migration by 38% (p = 0.01) and PC-3 migration by 25% (p = 0.02); whereas, a monoclonal anti-NGF antibody inhibited DU-145 migration by 40% (p = 0.01) and PC-3 migration by 37% (p = 0.02), at the same concentration. When IVIg was depleted of NGF-specific IgG by affinity chromatography, there was no significant inhibition of migration of the DU-145 and PC-3 cells at a concentration of 1 mg/well. Removal of the NGF-specific antibody from the IVIg was also demonstrated by a lack of effect on PC-12 cell differentiation. Therefore, IVIg is able to inhibit the migration of prostate cancer cell lines, through Matrigel chambers in vitro, only when the natural NGF-specific antibodies actively are present in IVIg.






Similar content being viewed by others
Abbreviations
- ECM:
-
Extracellular matrix
- IL-12:
-
Interleukin-12
- IVIg:
-
Intravenous gammaglobulin
- MEM:
-
Minimal essential medium
- NGF:
-
Nerve growth factor
- NTR:
-
Neurotrophin receptor
- OD:
-
Optical density
- TrKA:
-
Tyrosine kinase A
References
Jemal A, Siegel R, Ward E, Hao Y, Xu J, Thun MJ (2009) Cancer statistics, CA Cancer J Clin 59(4):225–249
Culig Z, Hobisch A, Cronauer MV et al (1996) Regulation of prostatic growth and function by peptide growth factors. Prostate 28(6):392–405
Bonfil RD, Chinni S, Fridman R, Kim HR, Cher ML (2007) Proteases, growth factors, chemokines, and the microenvironment in prostate cancer bone metastasis. Urol Oncol 25(5):407–411
Jacob K, Webber M, Benayahu D, Kleinman HK (1999) Osteonectin promotes prostate cancer cell migration and invasion: a possible mechanism for metastasis to bone. Cancer Res 59(17):4453–4457
Angelucci A, Festuccia C, D’Andrea G, Teti A, Bologna M (2002) Osteopontin modulates prostate carcinoma invasive capacity through RGD-dependent upregulation of plasminogen activators. Biol Chem 383(1):229–234
Hellawell GO, Turner GD, Davies DR, Poulsom R, Brewster SF, Macaulay VM (2002) Expression of the type 1 insulin-like growth factor receptor is up-regulated in primary prostate cancer and commonly persists in metastatic disease. Cancer Res 62(10):2942–2950
Heinlein CA, Chang C (2004) Androgen receptor in prostate cancer. Endocr Rev 25(2):276–308
Chinni SR, Sivalogan S, Dong Z, Filho JC, Deng X, Bonfil RD, Cher ML (2006) CXCL12/CXCR4 signaling activates Akt-1 and MMP-9 expression in prostate cancer cells: the role of bone microenvironment-associated CXCL12. Prostate 66(1):32–48
Luo Y, He DL, Ning L et al (2006) Over-expression of hypoxia-inducible factor-1alpha increases the invasive potency of LNCaP cells in vitro. BJU Int 98(6):1315–1319
Lambiase A, Micera A, Sgrulletta R, Bonini S, Bonini S (2004) Nerve growth factor and the immune system: old and new concepts in the cross-talk between immune and resident cells during pathophysiological conditions. Curr Opin Allergy Clin Immunol 4(5):425–430
Tometten M, Blois S, Arck PC (2005) Nerve growth factor in reproductive biology: link between the immune, endocrine and nervous system? Chem Immunol Allergy 89:135–148
Nockher WA, Renz H (2006) Neurotrophins in allergic diseases: from neuronal growth factors to intercellular signaling molecules. J Allergy Clin Immunol 117(3):583–589
Nockher WA, Renz H (2006) Neurotrophins and asthma: novel insight into neuroimmune interaction. J Allergy Clin Immunol 117(1):67–71
Linker R, Gold R, Luhder F (2009) Function of neurotrophic factors beyond the nervous system: inflammation and autoimmune demyelination. Crit Rev Immunol 29(1):43–68
Abram M, Wegmann M, Fokuhl V et al (2009) Nerve growth factor and neurotrophin-3 mediate survival of pulmonary plasma cells during the allergic airway inflammation. J Immunol 182(8):4705–4712
Krüttgen A, Schneider I, Weis J (2006) The dark side of the NGF family: neurotrophins in neoplasias. Brain Pathol 16(4):304–310
Papatsoris AG, Liolitsa D, Deliveliotis C (2007) Manipulation of the nerve growth factor network in prostate cancer. Expert Opin Investig Drugs 16(3):303–309 (Review)
Djakiew D, Delsite R, Pflug B, Wrathall J, Lynch JH, Onoda M (1991) Regulation of growth by a nerve growth factor-like protein which modulates paracrine interactions between a neoplastic epithelial cell line and stromal cells of the human prostate. Cancer Res 51(12):3304–3310
Delsite R, Djakiew D (1999) Characterization of nerve growth factor precursor protein expression by human prostate stromal cells: a role in selective neurotrophin stimulation of prostate epithelial cell growth. Prostate 41(1):39–48
Dalal R, Djakiew D (1997) Molecular characterization of neurotrophin expression and the corresponding tropomyosin receptor kinases (trks) in epithelial and stromal cells of the human prostate. Mol Cell Endocrinol 134(1):15–22
Pflug BR, Dionne C, Kaplan DR, Lynch J, Djakiew D (1995) Expression of a Trk high affinity nerve growth factor receptor in the human prostate. Endocrinology 136(1):262–268
Pflug BR, Onoda M, Lynch JH, Djakiew D (1992) Reduced expression of the low affinity nerve growth factor receptor in benign and malignant human prostate tissue and loss of expression in four human metastatic prostate tumor cell lines. Cancer Res 52(19):5403–5406
Geldof AA, De Kleijn MA, Rao BR, Newling DW (1997) Nerve growth factor stimulates in vitro invasive capacity of DU145 human prostatic cancer cells. J Cancer Res Clin Oncol 123(2):107–112
Walch ET, Marchetti D (1999) Role of neurotrophins and neurotrophins receptors in the in vitro invasion and heparanase production of human prostate cancer cells. Clin Exp Metastasis 17(4):307–314
Krygier S, Djakiew D (2001) Molecular characterization of the loss of p75(NTR) expression in human prostate tumor cells. Mol Carcinog 31(1):46–55
Krygier S, Djakiew D (2002) Neurotrophin receptor p75(NTR) suppresses growth and nerve growth factor-mediated metastasis of human prostate cancer cells. Int J Cancer 98(1):1–7
Festuccia C, Muzi P, Gravina GL et al (2007) Tyrosine kinase inhibitor CEP-701 blocks the NTRK1/NGF receptor and limits the invasive capability of prostate cancer cells in vitro. Int J Oncol 30(1):193–200
Warrington RJ, Lewis KE (2007) Biologically active anti-nerve growth factor antibodies in commercial intravenous gammaglobulin. J Autoimmun 28(1):24–29
Halvorson KG, Kubota K, Sevcik MA et al (2005) A blocking antibody to nerve growth factor attenuates skeletal pain induced by prostate tumor cells growing in bone. Cancer Res 65(20):9426–9435
Miknyoczki SJ, Wan W, Chang H et al (2002) The neurotrophin–trk receptor axes are critical for the growth, progression of human prostatic carcinoma, pancreatic ductal adenocarcinoma xenografts in nude mice. Clin Cancer Res 8(6):1924–1931
Schachter J, Katz U, Mahrer A et al (2007) Efficacy and safety of intravenous immunoglobulin in patients with metastatic melanoma. Ann N Y Acad Sci 1110:305–314
Damianovich M, Solomon AS, Blank M, Shoenfeld Y (2007) Attenuation of colon carcinoma tumor spread by intravenous immunoglobulin. Ann N Y Acad Sci 1110:567–577
Fishman P, Bar-Yehuda S, Shoenfeld Y (2002) IVIg to prevent tumor metastases. Int J Oncol 21(4):875–880
Muir D, Sukhu L, Johnson J, Lahorra MA, Maria BL (1993) Quantitative methods for scoring cell migration and invasion in filter-based assays. Anal Biochem 215(1):104–109
Saito K, Oku T, Ata N, Miyashiro H, Hattori M, Saiki I (1997) A modified and convenient method for assessing tumor cell invasion and migration and its application to screening inhibitors. Biol Pharm Bull 20(4):345–348
Wood P (2009) Primary antibody deficiency syndromes. Ann Clin Biochem 46(Pt 2):99–108
Darabi K, Abdel-Wahab O, Dzik WH (2006) Current usage of intravenous immune globulin and the rationale behind it: the Massachusetts General Hospital data and a review of the literature. Transfusion 46(5):741–753
Graff-Dubois S, Sibéril S, Elluru S et al (2007) Use of intravenous polyclonal immunoglobulins in autoimmune and inflammatory disorders. Transfus Clin Biol 14(1):63–68
Galeotti C, Maddur MS, Kazatchkine MD, Mouthon L, Kaveri SV (2009) Mechanisms of action of IVIG in autoimmune and inflammatory disorders: Recent developments. Transfus Clin Biol 16(2):75–79
Sarti L, Falai T, Pinto F, Tendi E, Matà S (2009) Intravenous immune globulin usage for neurological and neuromuscular disorders: an academic centre, 4 years experience. Neurol Sci 30(3):213–218
Wang X, Bauer JH, Li Y et al (2001) Characterization of a p75(NTR) apoptotic signaling pathway using a novel cellular model. J Biol Chem 276(36):33812–33820
Rabizadeh S, Bredesen DE (2003) Ten years on: mediation of cell death by the common neurotrophin receptor p75(NTR). Cytokine Growth Factor Rev 14(3–4):225–239
Khwaja F, Tabassum A, Allen J, Djakiew D (2006) The p75(NTR) tumor suppressor induces cell cycle arrest facilitating caspase mediated apoptosis in prostate tumor cells. Biochem Biophys Res Commun 341(4):1184–1192
Giraud S, Lautrette C, Bessette B, Decourt C, Mathonnet M, Jauberteau MO (2005) Modulation of Fas-induced apoptosis by p75 neurotrophin receptor in a human neuroblastoma cell line. Apoptosis 10(6):1271–1283
Weeraratna AT, Arnold JT, George DJ, DeMarzo A, Isaacs JT (2001) Rational basis for Trk inhibition therapy for prostate cancer. Prostate 45(2):140–148
Sigala S, Tognazzi N, Rizzetti MC, Farzoni I, Missale C, Bonmassar E, Spano P (2002) Nerve growth factor induces the re-expression of functional androgen receptors and p75(NGFR) in the androgen-insensitive prostate cancer cell line DU145. Eur J Endocrinol 147(3):407–415
Sigala S, Faraoni I, Botticini D, Paez-Pereda M, Missale C, Bonmassar E, Spano P (1999) Suppression of telomerase, re-expression of KA11, and abrogation of tumorigenicity by nerve growth factor in Prostate Cancer cell lines. Clin Cancer Res 5:1211–1218
Djakiew D, Pflug BR, Delsite R, Onoda M, Lynch JH, Arand G, Thompson EW (1993) Chemotaxis and chemokinesis of human prostate tumor cell lines in response to human prostate stromal cell secretory products containing a nerve growth factor-like protein. Cancer Res 53(6):1416–1420
Delsite R, Djakiew D (1996) Anti-proliferative effect of the kinase inhibitor K252a on human prostatic carcinoma cell lines. J Androl 17(5):481–490
Fraser FP, Salvador V, Manning EA, Mizal J, Alfun S, Raza M, Berridge RJ, Djamgoz MB (2003) Contribution of functional voltage-gated Na+ channel expression to cell behaviors involved in the metastatic cascade in rat cancer. J Cell Physiol 195(3):479–487
Diss JK, Stewart D, Pani F, Walker MM, Patel A, Djamgoz MB (2005) A potential novel marker for human prostate cancer: voltage-gated sodium channel expression in vivo. Prostate Cancer Prostatic Dis 8(3):266–273
Brackenbury WT, Djamgoz MB (2007) Nerve growth factor enhances voltage-gated Nat channel activity and transwell migration in Mat-LyLu rat prostate cancer cell line. J Cell Physiol 210(3):602–608
Acknowledgments
Funding for this work was provided by Bayer Inc. Canada.
Author information
Authors and Affiliations
Corresponding author
Additional information
Author Keith E. Lewis is deceased.
Rights and permissions
About this article
Cite this article
Warrington, R.J., Lewis, K.E. Natural antibodies against nerve growth factor inhibit in vitro prostate cancer cell metastasis. Cancer Immunol Immunother 60, 187–195 (2011). https://doi.org/10.1007/s00262-010-0934-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-010-0934-x