Abstract.
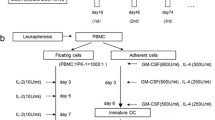
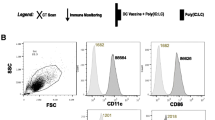
Dendritic cells (DC) derived from peripheral blood monocytes are currently being investigated in clinical trials for their role in stimulating the immune system. We performed a phase I/II clinical trial using human autologous DC transfected with cDNA of the human tumor antigen mucin (MUC1) as a vaccine in 10 patients with advanced breast, pancreatic or papillary cancer. After liposomal transfection, flow cytometry testing showed that 2% to 53% of the DC expressed mucin epitopes. Patients were immunized two or three times with 1 million transfected DC injected subcutaneously (s.c.). A vaccine-specific delayed-type hypersensitivity (DTH) reaction was observed in 3 out of 10 patients. After vaccination, 4 patients showed a 2- to 10-fold increase in the frequency of mucin-specific interferon-gamma (IFN-γ)-secreting CD8+ T cells. We demonstrated the feasibility and safety of a vaccine consisting of autologous gene-transfected DC, and that immunologic responses could be induced even in patients with pretreated and advanced disease.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Pecher, G., Häring, A., Kaiser, L. et al. Mucin gene (MUC1) transfected dendritic cells as vaccine: results of a phase I/II clinical trial. Cancer Immunol Immunother 51, 669–673 (2002). https://doi.org/10.1007/s00262-002-0317-z
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s00262-002-0317-z